No products in the cart.
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇫🇮 Finland
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇻 Latvia
🇱🇹 Lithuania
🇲🇽 Mexico
🇳🇱 Netherlands
🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇫🇮 Finland
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇻 Latvia
🇱🇹 Lithuania
🇲🇽 Mexico
🇳🇱 Netherlands
🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
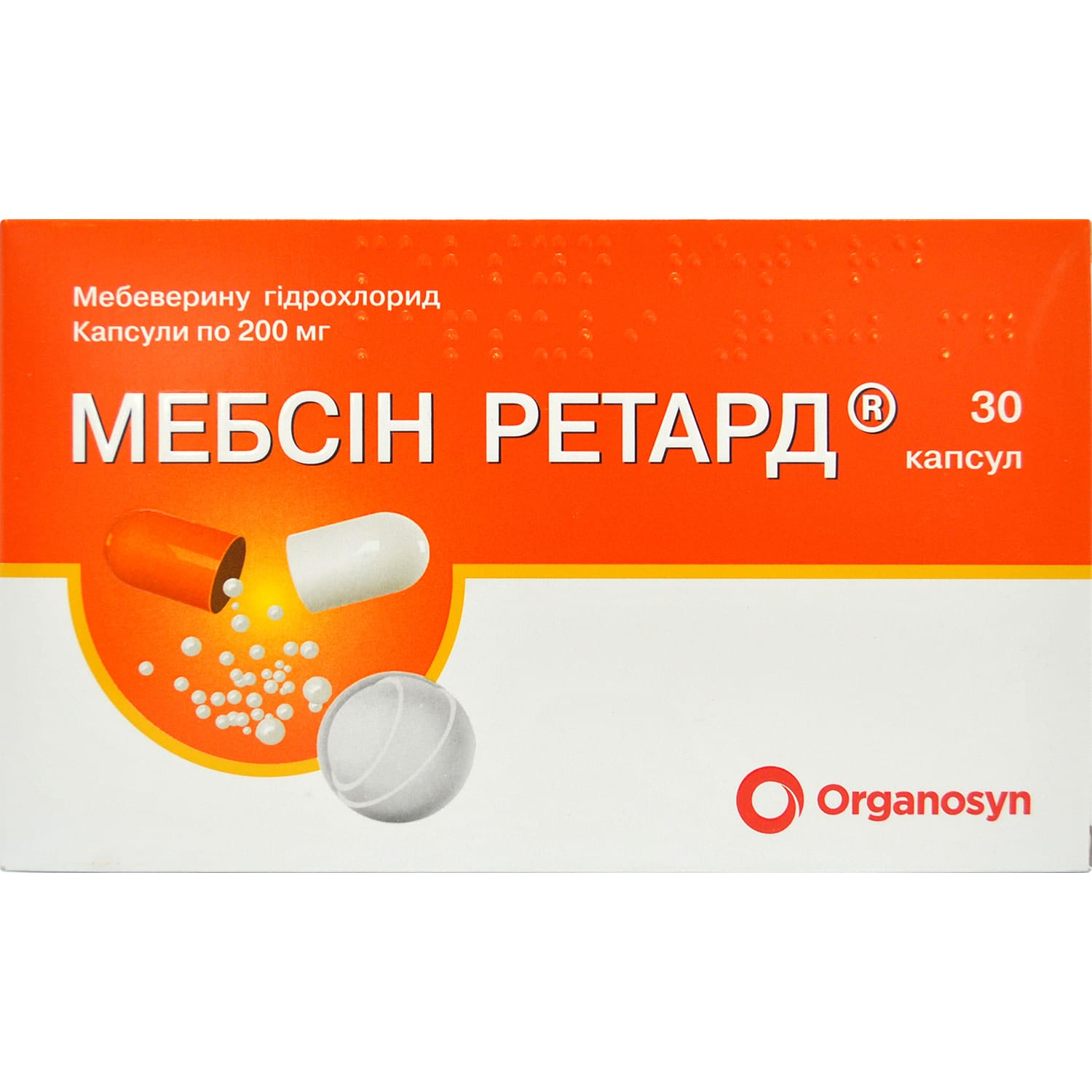
Mebsyn retard capsules 200 mg 30 pcs.
$25.10
Free Worldwide Shipping
to: Australia, Canada, Czechia, Denmark, Estonia, Finland, Ireland, Israel, Italy, Japan, Latvia, Lithuania, Mexico, Netherlands, Poland, Slovakia, South Korea, Switzerland, United Kingdom, United States and more
In stock
Mebsyn Retard capsules 200 mg 30 pcs relieve intestinal spasms, reduce IBS pain and discomfort, support normal gut motility with prolonged action.
Categories: Pain relief
Brand: Organosyn
Payment
PayPal, Debit or Credit card, Google Pay, Apple Pay
Composition and form of release
Composition
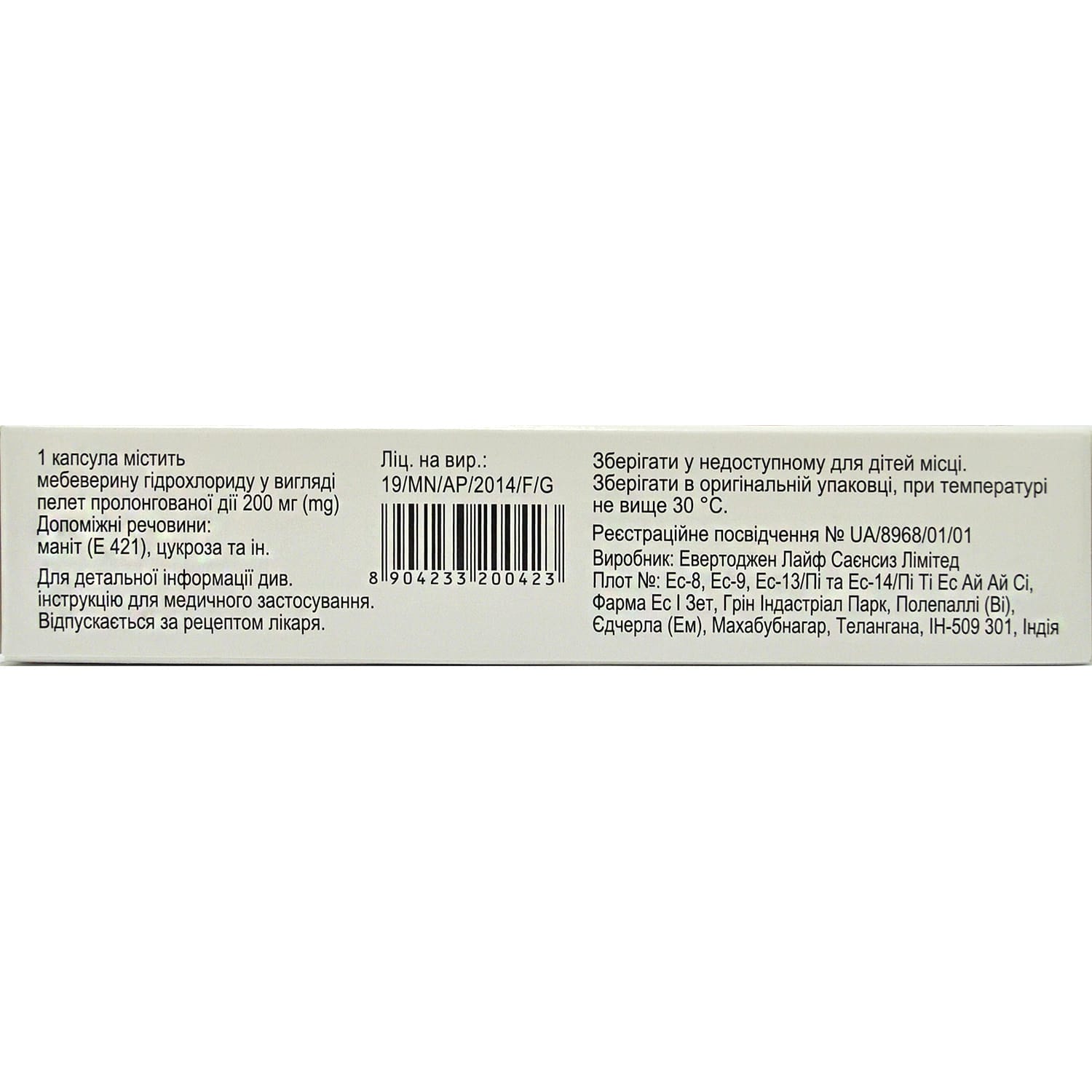
active ingredient: mebeverine hydrochloride (mebeverine);
1 capsule contains 200.0 mg of mebeverine hydrochloride in the form of prolonged-release pellets;
Excipients: table sugar, sucrose (sucrose), ethylcellulose, magnesium stearate, povidone, polyethylene glycol (macragol), hypromellose (hydroxypropyl methylcellulose).
Release form
Capsules.
Pharmacological properties
Pharmacodynamics. Mechanism of action and pharmacodynamic effects. Mebeverine is a myotropic antispasmodic with a selective effect on the smooth muscles of the digestive tract. It eliminates spasms without inhibiting normal intestinal motility. Since this action is not mediated by the autonomic nervous system, typical anticholinergic side effects do not occur.
Clinical efficacy and safety. The clinical efficacy and safety of different dosage forms of mebeverine have been studied in over 1500 patients. A significant reduction in the severity of the main symptoms of irritable bowel syndrome (e.g. abdominal pain, stool characteristics) was usually noted in reference and controlled clinical studies according to the main significance.
All dosage forms of mebeverine were generally safe and well tolerated at the recommended dosage regimen.
Children. Clinical studies with tablets or capsules have only been conducted in adults. Clinical efficacy and safety data, as well as post-marketing experience with mebeverine suspension in patients aged 3 years and older, have demonstrated that mebeverine is an effective, safe and well-tolerated drug.
Clinical trials of mebeverine suspension have shown it to be effective in alleviating symptoms of irritable bowel syndrome in children. Further open-label, placebo-controlled trials of mebeverine suspension have confirmed its efficacy.
The dosage regimen of tablets or capsules is calculated based on the safety and tolerability of mebeverine.
Pharmacokinetics. Absorption. Mebeverine is rapidly and completely absorbed after oral administration in tablet form. Due to the prolonged release of the drug from the capsule, it can be taken twice a day.
Distribution: With repeated use of Mebsyn Retard, no significant accumulation occurs.
Biotransformation. Mebeverine hydrochloride is mainly metabolized by esterases, which in the first stage of metabolism cleave the ester bonds with the formation of veratric acid and mebeverine alcohol. In blood plasma, demethylcarboxylic acid (DMCA) is the main metabolite. T ½ DMCA in the equilibrium state is 5.77 hours. With repeated use of capsules (200 mg 2 times a day), C max for DMCA was 804 ng / ml, and t max – about 3 hours. The relative bioavailability of prolonged-release capsules was optimal with an average ratio of 97%.
Excretion. Mebeverine is NOT excreted unchanged, it is completely metabolized, and the metabolites are excreted almost completely. Veratric acid is excreted in the urine. Mebeverine alcohol is also excreted by the kidneys as the carboxylic acid or DMCC.
Children: Pharmacokinetic studies in children have not been conducted.
Indication
Adults and children over 10 years of age:
- symptomatic treatment of abdominal pain and cramps, intestinal disorders and discomfort in the intestinal area in irritable bowel syndrome;
- treatment of gastrointestinal spasms of secondary origin caused by organic diseases.
Contraindication
Hypersensitivity to the active substance or any component of the drug.
Application
The drug is intended for oral use.
The capsules should be taken with sufficient water (at least 100 ml). It is not recommended to chew the capsule, as the capsule coating is designed for prolonged release of the drug.
Adults and children over 10 years of age should take 1 capsule 2 times a day (morning and evening).
The duration of use is not limited. If one or more doses are missed, the patient should take the next dose as prescribed. The missed dose should not be taken in addition to the next scheduled dose.
Special patient groups. No dosage studies have been conducted in elderly patients or patients with renal and/or hepatic impairment. Based on available post-marketing data, no specific risk has been identified for elderly patients or patients with renal and/or hepatic impairment. No dose adjustment is required for the above patient groups.
Special instructions
As the medicine contains sugar, patients with rare hereditary problems of galactose or fructose intolerance, the Lapp lactase deficiency, glucose-galactose malabsorption or sucrase-isomaltase insufficiency should not take this medicine.
Use during pregnancy and breastfeeding
The drug Mebsyn Retard is not recommended for use during pregnancy and breastfeeding.
Children
It is not prescribed to children under the age of 10 due to the high content of the active substance in the drug Mebsyn Retard.
Drivers
Studies on the effects on the ability to drive and use machines have not been conducted. The pharmacodynamic and pharmacokinetic profile, as well as post-marketing experience, do not indicate any harmful effects on the ability to drive and use machines.
Side effects
The following adverse reactions have been reported spontaneously during post-marketing surveillance. The frequency cannot be accurately estimated from the available data.
Allergic reactions have been identified, mainly on the skin.
Skin and subcutaneous tissue disorders: urticaria, angioedema, facial edema, hyperemia, skin rash, including hemorrhagic.
From the nervous system: headache, dizziness, depression.
Gastrointestinal tract: diarrhea or constipation.
General disorders: hypersensitivity (anaphylactic reactions).
Overdose
In case of overdose, theoretically, central nervous system disorders may occur. In cases of mebeverine overdose, symptoms were absent or mild and disappeared quickly. Symptoms of overdose that occurred were of neurological or cardiovascular origin.
Treatment. No specific antidote is known. Symptomatic treatment is recommended. Gastric lavage is recommended only in case of intoxication with several different drugs, which is diagnosed within 1 hour of taking the drugs. Measures to reduce absorption are not necessary.
Interactions
Interaction studies were conducted only with alcohol. In vitro and in vivo studies demonstrated the absence of any interaction of the drug and ethanol.
Storage conditions
In the original packaging at a temperature not exceeding 30 °C.
Be the first to review “Mebsyn retard capsules 200 mg 30 pcs.” Cancel reply
You may also like








Reviews
There are no reviews yet.