No products in the cart.
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇫🇮 Finland
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇻 Latvia
🇱🇹 Lithuania
🇲🇽 Mexico
🇳🇱 Netherlands
🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇫🇮 Finland
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇻 Latvia
🇱🇹 Lithuania
🇲🇽 Mexico
🇳🇱 Netherlands
🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
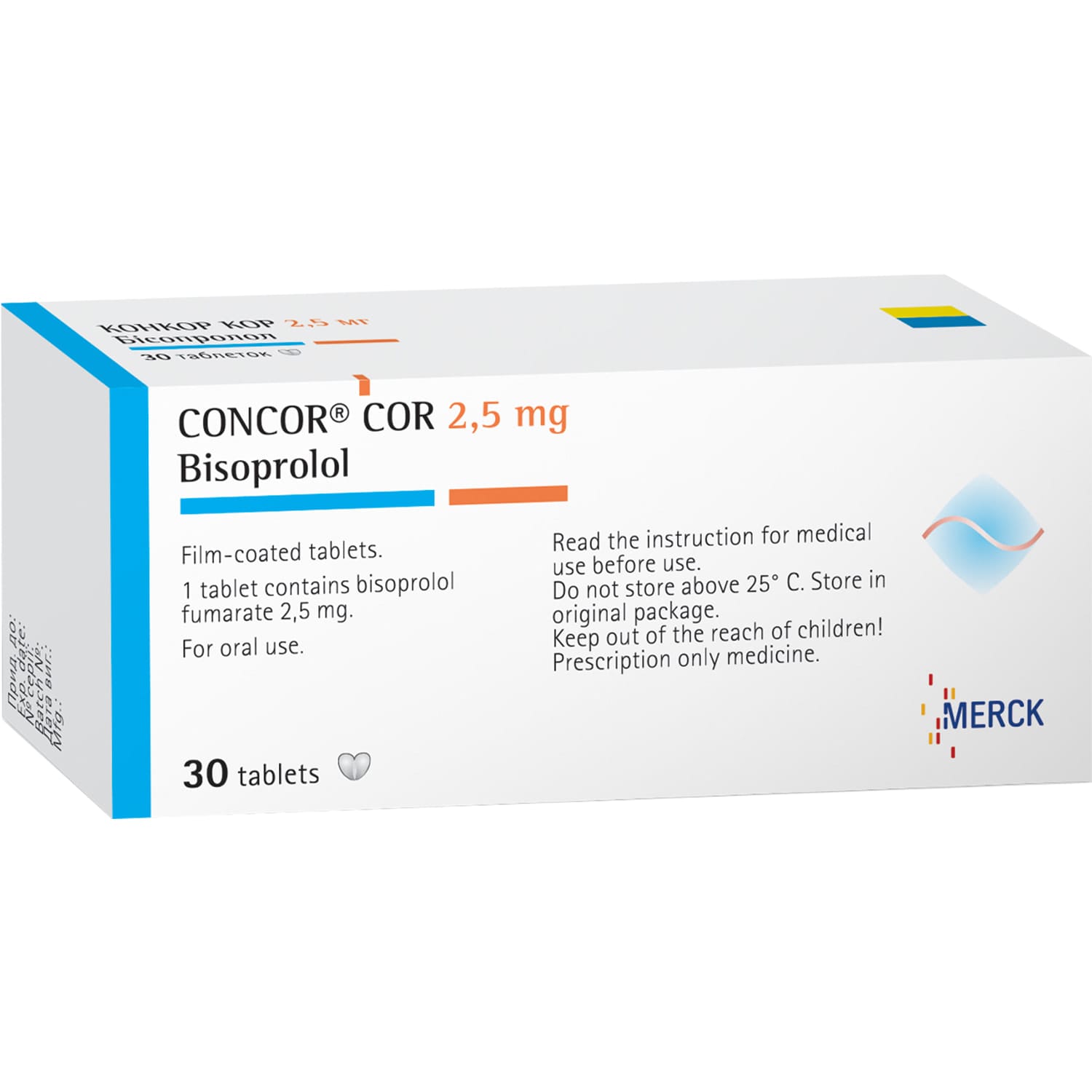
Concor Cor film-coated tablets 2.5 mg 30 pcs.
$23.20
Free Worldwide Shipping
to: Australia, Canada, Czechia, Denmark, Estonia, Finland, Ireland, Israel, Italy, Japan, Latvia, Lithuania, Mexico, Netherlands, Poland, Slovakia, South Korea, Switzerland, United Kingdom, United States and more
In stock
Concor Cor is a selective β1-blocker used in chronic heart failure to reduce heart rate, blood pressure, and cardiac workload.
Categories: Heart and blood vessels, Hematopoiesis and blood
Brand: Merck
Payment
PayPal, Debit or Credit card, Google Pay, Apple Pay
Pharmacological properties
Pharmacodynamics. The active substance of the drug Concor cor – bisoprolol – is a selective β1-adrenoreceptor blocker. When used in therapeutic doses, it does not have intrinsic sympathomimetic activity and clinically significant membrane-stabilizing properties. It has antianginal and hypotensive effects. Concor cor reduces myocardial oxygen demand by reducing heart rate, cardiac output and blood pressure, increases myocardial oxygen supply by reducing end-diastolic blood pressure and prolonging diastole. The drug has a very low affinity for β2-receptors of smooth muscles of the bronchi and blood vessels, as well as β2-receptors of the endocrine system. The drug can only in rare cases affect the smooth muscles of the bronchi and peripheral arteries, as well as glucose metabolism.
Pharmacokinetics. Absorption and distribution. After oral administration, Concor Cor is well absorbed in the gastrointestinal tract. Bioavailability is about 90% and does not depend on food intake. The volume of distribution is 3.5 l/kg of body weight. The total clearance of bisoprolol is 15 l/h. Due to the long T ½ (10-12 h), the drug retains its therapeutic effect for 24 hours after a single dose.
Metabolism and excretion. Bisoprolol is excreted from the body in two ways: 50% is metabolized in the liver with the formation of inactive metabolites and excreted through the kidneys, 50% is excreted in the urine unchanged.
The pharmacokinetics of bisoprolol are linear and independent of age.
Indication
Treatment of chronic heart failure with left ventricular systolic dysfunction in combination with ACE inhibitors, diuretics, and, if necessary, cardiac glycosides.
Application
Concor cor should be taken without chewing, in the morning on an empty stomach or during breakfast, with a small amount of liquid.
Standard therapy for chronic heart failure: ACE inhibitors or angiotensin II antagonists, β-adrenergic blockers, diuretics, and, if necessary, cardiac glycosides.
At the beginning of treatment with bisoprolol, the patient should not have signs of exacerbation. Transient worsening of heart failure, hypotension or bradycardia may occur during and after the titration period.
Dose titration period. Treatment of chronic heart failure with bisoprolol begins in the following titration order and can be adjusted depending on individual body reactions:
- 1.25 mg bisoprolol fumarate once daily for 1 week, if well tolerated, increase to
- 2.5 mg bisoprolol fumarate once daily for the next 1 week, if well tolerated, increase to
- 3.75 mg bisoprolol fumarate once daily for the next 1 week, if well tolerated, increase to
- 5 mg bisoprolol fumarate once daily for the next 4 weeks, if well tolerated, increase to
- 7.5 mg bisoprolol fumarate once daily for the next 4 weeks, if well tolerated, increase to
- 10 mg bisoprolol fumarate once daily as maintenance therapy.
The maximum recommended dose of bisoprolol hemifumarate is 10 mg once daily.
During the titration phase, vital signs (BP, HR) and symptoms of heart failure progression should be closely monitored. Symptoms may occur from the first day after initiation of treatment.
Treatment modification. If worsening of heart failure, hypotension or bradycardia develops during or after the titration phase, it is recommended to adjust the dose of the drug, which may require a temporary reduction in the dose of bisoprolol or discontinuation of treatment. After stabilization of the patient’s condition, treatment with the drug is continued.
The course of treatment with Concor Cor is long.
Do not stop treatment suddenly or change the recommended dose without consulting your doctor, as this may worsen the patient’s condition. If necessary, treatment with the drug should be discontinued slowly, gradually reducing the dose.
Patients with hepatic and renal insufficiency. There are no data on the pharmacokinetics of bisoprolol in patients with chronic heart failure concomitant with impaired hepatic and/or renal function, therefore, the dose should be increased with extreme caution.
Elderly patients: No dose adjustment is required.
Contraindication
Acute heart failure or heart failure in a state of decompensation, requiring intravenous inotropic therapy; cardiogenic shock; AV block II and III degree; sick sinus syndrome; severe sinoatrial block; symptomatic bradycardia; symptomatic arterial hypotension; severe form of BA; severe form of obliterating peripheral arterial diseases or Raynaud’s disease; untreated pheochromocytoma; metabolic acidosis; hypersensitivity to bisoprolol or to other components of the drug.
Side effects
Adverse effects are classified by frequency of occurrence into the following categories: very common (≥1/10), common (≥1/100 and 1/10), uncommon (≥1/1000 and 1/100), rare (≥1/10,000 and 1/1000), very rare (1/10,000).
From the cardiovascular system: very often – bradycardia; often – signs of worsening heart failure, feeling of coldness or numbness of the extremities, arterial hypotension, especially in patients with heart failure; infrequently – AV conduction disorders, orthostatic hypotension.
From the nervous system: often – dizziness, headache; rarely – syncope.
On the part of the organ of vision: rarely – decreased tear secretion (should be taken into account when wearing contact lenses); very rarely – conjunctivitis.
On the part of the organ of hearing: rarely – hearing impairment.
On the part of the respiratory system: infrequently – bronchospasm in patients with a history of bronchial asthma and chronic obstructive respiratory diseases; rarely – allergic rhinitis.
Gastrointestinal tract: often – nausea, vomiting, diarrhea, constipation.
On the part of the skin: rarely – hypersensitivity reactions (itching, redness, rash); very rarely – when treated with β-adrenergic blockers, the condition of patients with psoriasis may worsen in the form of psoriatic rash, alopecia.
Musculoskeletal system: infrequently – muscle weakness, cramps.
On the part of the liver: rarely – hepatitis.
From the reproductive system: rarely – impaired potency.
Mental disorders: infrequently – depression, sleep disorders; rarely – nightmares, hallucinations.
Laboratory indicators: rarely – increased TG levels in the blood, increased activity of liver enzymes in blood plasma (AST, ALT).
The body as a whole: often – asthenia, fatigue.
In case of side effects or adverse reactions, you should immediately inform your doctor.
Special instructions
Treatment of stable chronic heart failure with bisoprolol should begin with a titration phase.
In patients with coronary artery disease, treatment should not be stopped abruptly without necessity, as this may lead to transient deterioration of the condition. Initiation and discontinuation of bisoprolol treatment requires regular monitoring.
Currently, there is insufficient therapeutic experience in the treatment of heart failure in patients with the following diseases and pathological conditions: type 1 diabetes mellitus (insulin-dependent), severe renal dysfunction, severe hepatic dysfunction, restrictive cardiomyopathy, congenital heart disease, hemodynamically significant acquired valvular heart disease, myocardial infarction within the last 3 months.
The drug should be used with caution in patients with the following conditions:
- bronchospasm (with asthma or COPD);
- diabetes with sharp fluctuations in blood glucose levels; symptoms of hypoglycemia may be hidden;
- strict diet;
- during desensitization therapy. Like other β-adrenergic blockers, bisoprolol may increase sensitivity to allergens and increase the severity of anaphylactic reactions. In such cases, treatment with adrenaline does not always give a positive therapeutic effect;
- First degree AV block;
- Prinzmetal’s angina;
- Obliterating peripheral arterial diseases (at the beginning of therapy, complaints may worsen);
- general anesthesia.
It is necessary to warn the anesthesiologist about taking β-adrenergic blockers. In patients who are scheduled for general anesthesia, the use of β-adrenergic blockers reduces the risk of arrhythmia and myocardial ischemia during induction of anesthesia, intubation and the postoperative period. It is recommended to continue the use of β-adrenergic blockers in the perioperative period. The anesthesiologist should take into account the potential interaction with other drugs, which can lead to bradyarrhythmia, reflex tachycardia and a decrease in the ability of the reflex mechanism to compensate for blood loss. If bisoprolol is discontinued before surgery, the dose should be gradually reduced and the drug should be discontinued 48 hours before general anesthesia.
Combinations of bisoprolol with calcium antagonists of the verapamil or diltiazem group, with class I antiarrhythmic drugs and with centrally acting antihypertensive agents are not recommended (see Interaction with other drugs).
Although cardioselective β-adrenergic (β 1) blockers have less effect on lung function than non-selective β-blockers, their use, like all β-adrenergic blockers, should be avoided in obstructive airway diseases unless there are compelling reasons for therapy. If necessary, Concor Cor should be used with caution. In patients with obstructive airway diseases, treatment with bisoprolol should be started at the lowest possible dose and patients should be monitored for the appearance of new symptoms (such as shortness of breath, exercise intolerance, cough).
In patients with asthma or other COPD conditions that may cause symptoms, concomitant bronchodilator therapy is indicated. In some cases, higher doses of β2 -sympathomimetics may be required in patients with asthma due to increased airway resistance while taking the drug .
Patients with psoriasis (including those with a history of psoriasis) are prescribed β-adrenergic blockers (e.g. bisoprolol) after careful consideration of the benefit/risk ratio.
Patients with pheochromocytoma are prescribed bisoprolol only after the appointment of therapy with α-adrenergic blockers. Symptoms of thyrotoxicosis may be masked against the background of taking the drug.
Use during pregnancy or breastfeeding. During pregnancy, the drug is used only when the expected benefit to the mother outweighs the potential risk to the fetus. As a rule, α-blockers reduce blood flow in the placenta and may affect fetal development. It is necessary to monitor uteroplacental blood flow.
After delivery, the newborn should be closely monitored. Symptoms of hypoglycemia and bradycardia can be expected to develop during the first 3 days.
There is no data on the excretion of bisoprolol into breast milk or the safety of effects on breastfed infants, therefore Concor Cor is not recommended for use during breastfeeding.
Children. Clinical data on the efficacy and safety of the drug in children are lacking, therefore the drug should not be used in this category of patients.
Ability to influence the reaction rate when driving vehicles or working with other mechanisms. During studies involving patients with coronary artery disease, the drug did not affect the ability to drive vehicles. However, in some cases, the drug may affect the ability to drive vehicles or work with complex mechanisms. Special attention should be paid at the beginning of treatment, when changing the dose of the drug or when interacting with alcohol.
Interactions
Combinations that are not recommended for use
Treatment of chronic heart failure. Class I antiarrhythmics (e.g. quinidine, disopyramide, lidocaine, phenytoin, flecainide, propafenone): possible potentiation of the effect on AV conduction and increased severity of the negative inotropic effect.
All indications. Calcium antagonists such as verapamil, to a lesser extent diltiazem: negative effect on myocardial contractile function and AV conduction. Intravenous administration of verapamil can lead to severe hypotension and AV block.
Centrally acting antihypertensives (clonidine, methyldopa, moxonidine, rilmenidine): possible worsening of heart failure due to decreased central sympathetic tone (decreased heart rate and cardiac output, vasodilation). Abrupt withdrawal of the drug, especially if preceded by withdrawal of β-adrenergic blockers, may increase the risk of rebound hypertension.
Combinations to be used with caution
Treatment of hypertension or coronary artery disease (angina pectoris). Class I antiarrhythmics (e.g. quinidine, disopyramide, lidocaine, phenytoin, flecainide, propafenone): possible potentiation of the effect on AV conduction and increased severity of the negative inotropic effect.
All indications. Dihydropyridine calcium antagonists (e.g. nifedipine, felodipine, amlodipine) may increase the risk of hypotension. The possibility of an increased negative effect on myocardial inotropic function in patients with heart failure cannot be excluded.
Class III antiarrhythmic drugs (e.g. amiodarone) may increase the negative effect on AV conduction.
Topical β-adrenergic blockers (e.g. contained in eye drops for the treatment of glaucoma): possible enhancement of the systemic effects of bisoprolol.
Parasympathomimetics: AV conduction time may be increased and the risk of bradycardia may increase.
Insulin and oral antidiabetic agents: increased hypoglycemic effect. Blockade of β-adrenergic receptors may mask the symptoms of hypoglycemia.
Anesthetics: increased risk of myocardial depression and hypotension (see Precautions).
Cardiac glycosides: decrease in heart rate, increase in AV conduction time. NSAIDs may weaken the hypotensive effect of bisoprolol.
β-Sympathomimetics (e.g. orciprenaline, isoprenaline, dobutamine): use in combination with Concor Cor may lead to a decrease in the therapeutic effect of both drugs. For the treatment of allergic reactions, the use of higher doses of adrenaline may be required.
Sympathomimetics that activate α- and β-adrenergic receptors (e.g. adrenaline, noradrenaline): α-adrenergic mediated vasoconstriction may occur, leading to increased blood pressure and increased intermittent claudication. This interaction is more likely with non-selective β-adrenergic blockers.
When used simultaneously with antihypertensive agents and agents that have a hypotensive effect (e.g. tricyclic antidepressants, barbiturates, phenothiazines), the risk of hypotension may be increased.
Possible combinations: Mefloquine may increase the risk of bradycardia.
MAO inhibitors (except MAO inhibitors type B) enhance the hypotensive effect of β-adrenergic blockers, but there is a risk of developing hypertensive crisis.
Overdose
Symptoms. In case of overdose (for example, a daily dose of 15 mg instead of 7.5 mg), cases of third-degree AV block, bradycardia and dizziness have been recorded. Frequent signs of overdose of β-adrenergic blockers are bradycardia, hypotension, acute heart failure, hypoglycemia and bronchospasm. Several cases of overdose in patients with AG and/or coronary artery disease (maximum dose – 2000 mg of bisoprolol) are currently known. Bradycardia and/or hypotension were noted. All patients recovered. There is a wide variability in individual sensitivity to a single high dose of bisoprolol, patients with heart failure may be more sensitive to the drug. Therefore, treatment should be started with a gradual increase in dose (see application).
Treatment. In case of overdose, discontinue treatment with the drug and provide supportive and symptomatic therapy. There is limited evidence that bisoprolol is difficult to dialyze. If overdose is suspected, the following general measures should be considered, based on the expected pharmacological effect and recommendations for other β-adrenergic blockers. For bradycardia: intravenous administration of atropine. If there is no response, isoprenaline or another drug with a positive chronotropic effect should be administered with caution. In exceptional cases, transvenous administration of an artificial pacemaker may be required.
For hypotension: IV fluids and vasoconstrictors. IV glucagon may be useful.
For AV block II and III degree: careful observation and infusion of isoprenaline or transvenous introduction of a pacemaker.
In case of exacerbation of chronic heart failure: administration of diuretics, inotropic drugs, vasodilators.
For bronchospasm: bronchodilators (e.g. isoprenaline), β2 -adrenomimetics and/or aminophylline.
For hypoglycemia: intravenous glucose administration.
Storage conditions
At a temperature not exceeding 25 °C.
Be the first to review “Concor Cor film-coated tablets 2.5 mg 30 pcs.” Cancel reply
You may also like




Reviews
There are no reviews yet.