No products in the cart.
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
Menovazan ointment tube 40 g
$18.33


Benzyl benzoate skin emulsion ...
$19.79 Original price was: $19.79.$17.99Current price is: $17.99.
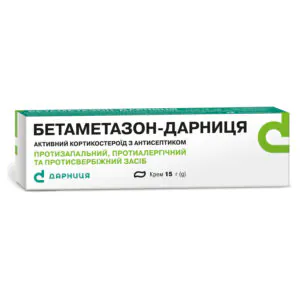
Betamethasone Darnytsia cream tube 15 g
$15.20
Free Worldwide Shipping
to: Australia, Canada, Czechia, Denmark, Estonia, Ireland, Israel, Italy, Japan, Lithuania, Mexico, Netherlands, Poland, Slovakia, South Korea, Switzerland, United Kingdom, United States and more
In stock
Betamethasone Darnytsia cream relieves inflammation, itching and allergic skin reactions in dermatitis, eczema and psoriasis with antibacterial support.
Categories: Dermatology
Brand: Darnytsia
Payment
PayPal, Debit or Credit card, Google Pay, Apple Pay
Pharmacological properties
Betamethasone valerate, which is part of the drug, has a local anti-inflammatory, anti-allergic, antipruritic and anti-exudative effect.
Cetylpyridinium chloride has a bacteriostatic and fungistatic effect, as a result of which the drug prevents the development of secondary infection.
Indication
Reducing the severity of inflammatory manifestations of dermatoses sensitive to corticosteroid therapy, such as eczema (atopic, nummular), neurodermatitis, dermatitis (contact, solar, seborrheic, exfoliative, radiation, intertriginous, stasis dermatitis), psoriasis (except widespread psoriasis), anogenital and senile itching.
Application
The drug is used externally. The cream is applied in a thin layer to the affected skin surface 1-3 times a day, depending on the severity of the disease. The frequency of application depends on the degree of damage: with a mild degree, the drug can be applied once a day, in case of more severe lesions, the frequency of application of the drug must be increased. In most cases, to achieve the effect, it is enough to apply the drug 1-2 times a day.
Do not use the drug under occlusive dressings, as the side effects of the drug may be increased.
Continuous use of the drug should not exceed 4 weeks. If the condition does not improve or worsens within 2-4 weeks of therapy, a re-examination should be performed and the treatment reviewed.
Contraindication
Hypersensitivity to the active substances or other components of the drug; bacterial, viral, fungal skin infections (including herpes simplex and herpes zoster, chickenpox, tuberculosis and syphilis of the skin), post-vaccination skin reactions, rosacea, rosacea-like (perioral) dermatitis.
The drug is not intended for use in ophthalmology.
Side effects
On the part of the immune system, skin and subcutaneous tissue: hypersensitivity reactions, burning and / or tingling of the skin, feeling of warmth, itching, irritation, dryness, cracking, lamellar and / or focal peeling of the skin, erythema, telangiectasia, folliculitis, follicular rash, acne-like rash, hypertrichosis, sweating, hypopigmentation, rosacea-like (perioral) dermatitis, allergic contact dermatitis, skin induration, skin maceration, skin atrophy, striae, secondary infection.
On the part of the endocrine system: suppression of the hypothalamic-pituitary-adrenal system (HPA) with the development of secondary adrenal insufficiency, symptoms of hypercorticism, Itsenko-Cushing’s syndrome.
Any adverse reactions seen with systemic use of corticosteroids, including suppression of adrenal cortex function, may also occur with their topical use, especially in high doses, for prolonged periods, under occlusive dressings, and in children.
In children receiving topical corticosteroids, cases of adrenal suppression, Cushing’s syndrome, growth retardation, failure to gain weight, and intracranial hypertension have been reported. Symptoms of adrenal suppression in children include low plasma and urinary cortisol levels and unresponsiveness to ACTH stimulation. Manifestations of intracranial hypertension include fontanelle swelling, headache, and bilateral optic nerve edema.
Special instructions
The drug is not intended for use in ophthalmology (on the mucous membrane of the eye, around the eyes).
Prolonged use of the drug on the skin of the face should be avoided.
Caution should be exercised when applying the drug near the face and avoid contact with the eyes and mucous membranes.
The drug is not recommended for use under occlusive dressings and/or on large areas of skin (high doses) and/or for a long time (more than 3-4 weeks) due to systemic absorption of betamethasone.
Systemic absorption of local corticosteroids increases with their dose. Highly active corticosteroids on large areas of skin should be used under the supervision of a physician and with periodic monitoring of the patient’s condition, since they can cause suppression of the GHNS. GHNS function is usually restored when the drug is discontinued. In some cases, withdrawal symptoms may develop, requiring the addition of systemic corticosteroids. In the event of the development of GHNS suppression, the frequency of application should be reduced or the drug should be discontinued and the patient transferred to a less potent GHNS.
If skin irritation or signs of hypersensitivity occur, discontinue use and consult a doctor.
In the event of fungal or bacterial superinfection of the skin, additional use of an antifungal or antibacterial agent is necessary. If the desired effect does not occur quickly, the use of corticosteroids should be discontinued until signs of infection resolve.
Use during pregnancy and breastfeeding. The drug should not be used in the first trimester of pregnancy.
The drug should only be prescribed in later stages of pregnancy and when the expected benefit to the mother clearly outweighs the potential risk to the fetus. Drugs of this group should not be used in pregnant women in high doses, for a long time, or under occlusive dressings.
It is not currently known whether topical corticosteroids, due to systemic absorption, can lead to their penetration into breast milk, therefore, when deciding whether to discontinue breast-feeding or discontinue the drug, the importance of the drug to the mother should be taken into account.
Children. The safety of the drug in children has not been established, therefore the drug is not intended for use in this category of patients.
Ability to influence the reaction rate when driving vehicles or other mechanisms. Usually the drug does not affect the patient’s reaction rate when driving vehicles or other mechanisms.
Interactions
There are no data on interactions with other drugs or other types of interactions.
However, the drug is not recommended for use simultaneously with topical forms of drugs, in particular those containing anionic surfactants that inactivate the effect of cetylpyridinium chloride.
Due to the presence of mineral oil in the excipients, the use of the drug in the anogenital area may damage the structure of latex condoms and reduce their safety during use.
In cases where systemic absorption of betamethasone can be assumed, one should be aware of numerous interactions with other drugs.
Overdose
Symptoms. Excessive or prolonged use of topical corticosteroids may cause suppression of pituitary-adrenal function, leading to secondary adrenal insufficiency and the appearance of symptoms of hypercorticism, including Itsenko-Cushing’s syndrome. Acute symptoms of hypercorticism are usually reversible.
Treatment: symptomatic therapy, if necessary, correction of electrolyte balance should be carried out. In case of chronic toxic effects, gradual withdrawal of the drug is recommended.
Storage conditions
At a temperature not exceeding 25 °C.
Be the first to review “Betamethasone Darnytsia cream tube 15 g” Cancel reply
You may also like










Reviews
There are no reviews yet.