No products in the cart.
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more

AEvit (vitamins A+E) soft caps...
$30.09 Original price was: $30.09.$18.99Current price is: $18.99.
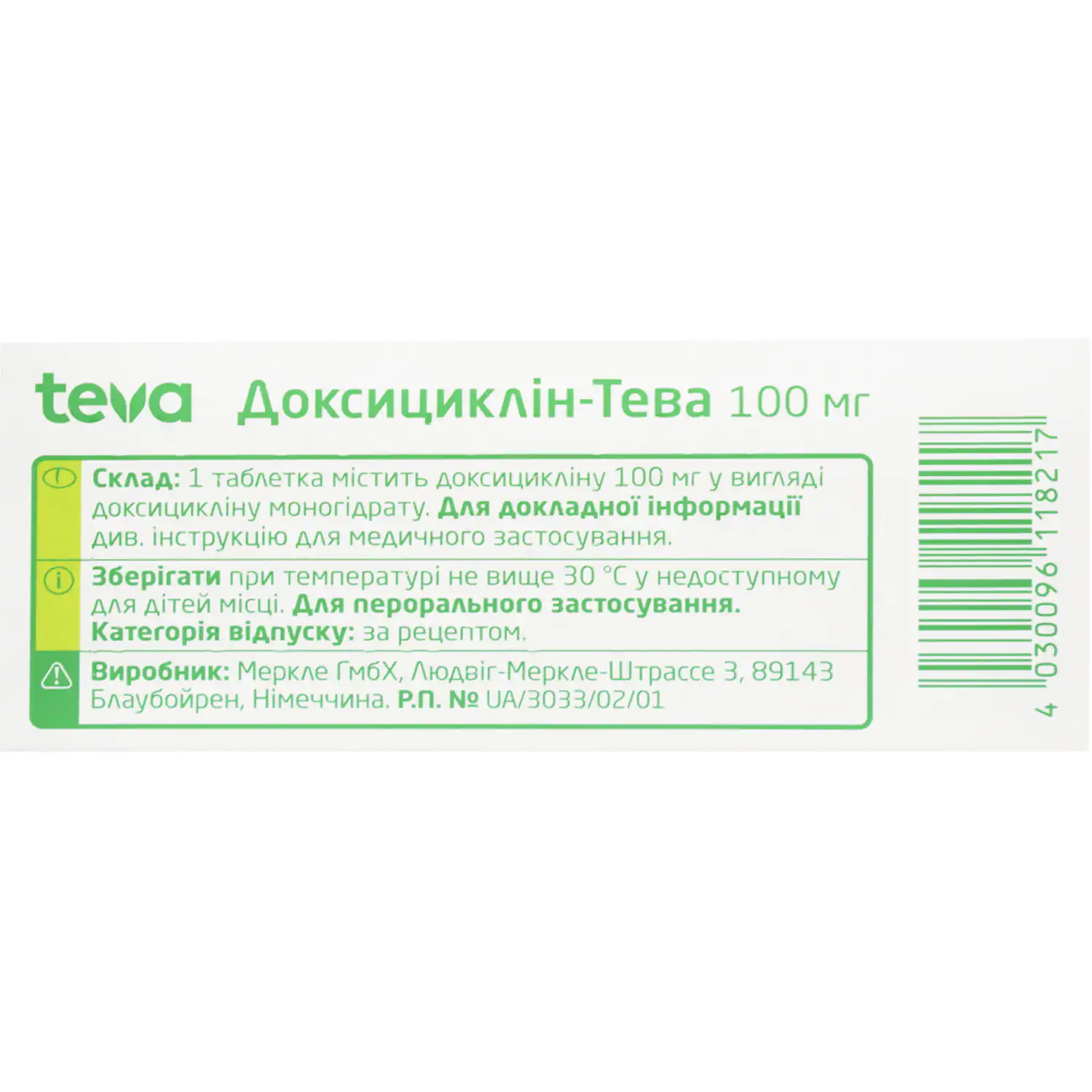
Doxycycline Teva tablets 100mg blister 10 pcs
$19.63
Free Worldwide Shipping
to: Australia, Canada, Czechia, Denmark, Estonia, Ireland, Israel, Italy, Japan, Lithuania, Mexico, Netherlands, Poland, Slovakia, South Korea, Switzerland, United Kingdom, United States and more
In stock
Doxycycline is a broad-spectrum antibiotic used to treat respiratory, urinary, skin infections, acne, and STIs. Learn dosage, side effects, and precautions.
Categories: Antimicrobials, Vitamins and supplements
Brand: Teva
Payment
PayPal, Debit or Credit card, Google Pay, Apple Pay
Pharmacological properties
Pharmacodynamics
Doxycycline has a bacteriostatic effect; its antimicrobial effect is realized by inhibiting protein synthesis. The drug is effective against a wide range of gram-positive and gram-negative bacteria and other microorganisms.
Doxycycline is used to treat infections caused by the following microorganisms: Staphylococcus, Streptococcus, Pneumococcus, Salmonella typhi, Klebsiella, Morganella morganii, Escherichia coli, Haemophilus influenzae, Clostridium, Bacteroides, Fusobacterium, Legionella pneumophila. Particular activity has been noted against the following microorganisms: Brucella, Pasteurella, Chlamydia, Mycoplasma pneumoniae, Rickettsia, Neisseria gonorrhoeae, Treponema, Spirocheta, Vibrio cholerae, Corynebacterium acnes.
Pharmacokinetics
Doxycycline is almost completely absorbed after oral administration. Studies show that the absorption of doxycycline differs from that of other tetracyclines. Simultaneous food intake does not significantly affect the absorption of the drug, however, dairy products significantly reduce its absorption due to the calcium content in the composition and the formation of poorly soluble chelate complexes.
After a dose of 200 mg, C max of doxycycline in the blood plasma of healthy adult volunteers averaged 2.6 μg/ml after 2 hours and decreased to 1.45 μg/ml after 24 hours. T ½ is 15-22 hours and is not significantly dependent on renal function.
Tetracyclines are easily absorbed and are 85-96% bound to blood plasma proteins. They are well distributed in tissues and body fluids, accumulating in high concentrations in bile and saliva, as well as in pleural, ascitic and synovial fluids. Therapeutic concentrations are maintained in the lungs, ovaries, prostate, testicles and liver. The drug also accumulates in bones and teeth. Doxycycline penetrates the placental barrier and into breast milk. A small amount of the drug penetrates the cerebrospinal fluid, but this is of no clinical significance. Doxycycline is metabolized in the liver and partially reabsorbed in the intestine. Unlike other tetracyclines, doxycycline is mainly (up to 70%) excreted in the feces, partly in the urine.
Indication
Doxycycline-Teva is used to treat various infections caused by sensitive strains of gram-positive and gram-negative microorganisms, as well as some other microorganisms.
Respiratory tract infections: pneumonia and other lower respiratory tract diseases caused by susceptible strains of Streptococcus pneumoniae, Haemophilus influenza, Klebsiella pneumoniae. Pneumonia caused by Mycoplasma pneumoniae. Treatment of chronic bronchitis, sinusitis.
Urinary tract infections: infections caused by sensitive strains of the species Klebsiella, Enterobacter, as well as Escherichia coli, Streptococcus faecalis.
Sexually transmitted diseases: infections caused by Chlamydia trachomatis, including uncomplicated urethral and endocervical infections and rectal infections. Non-gonococcal urethritis caused by Ureaplasma urealyticum (T-mycoplasma). Soft chancre, inguinal granuloma, venereal granuloma. Doxycycline is an alternative drug for the treatment of gonorrhea and syphilis.
Skin infections: acne if antibiotic therapy is necessary.
Since doxycycline belongs to the group of tetracycline antibiotics, it can be used for infections caused by microorganisms sensitive to tetracycline, namely:
Ophthalmic infections: infections caused by susceptible bacteria gonococci, staphylococci and Haemophilus influenza. The infection that causes trachoma is not always eliminated, which is confirmed by immunofluorescence analysis. For the treatment of paratrachoma, doxycycline can be used as monotherapy or in combination with other drugs.
Rickettsial infections: Rocky Mountain spotted fever, typhus group, Q fever, endocarditis caused by Coxiella, tick-borne fever.
Other infections: ornithosis, brucellosis (when used in combination with streptomycin), cholera, bubonic plague, epidemic relapsing typhus; tick-borne relapsing fever; tularemia, melioidosis, chloroquine-resistant tropical malaria, and acute intestinal amebiasis (when used in combination with an amebicide).
Doxycycline is an alternative drug for the treatment of leptospirosis, gas gangrene, and tetanus.
Doxycycline is indicated for the prevention of the following conditions: Japanese river fever, traveler’s diarrhea (caused by enterotoxigenic Escherichia coli), leptospirosis, malaria. Malaria prophylaxis should be administered in accordance with current practice due to the possibility of resistance development.
Application
The drug should be taken during meals, with plenty of liquid (NOT milk or dairy products), in an upright position. It is not recommended to lie down immediately after taking the drug.
Adults. The usual dose of doxycycline in adults for the treatment of acute infections is 200 mg on the 1st day of treatment (as a single dose or 100 mg every 12 hours) and 100 mg/day on subsequent days. In severe infections, the drug should be used at a dose of 200 mg/day throughout the entire treatment period.
Exceeding the recommended dose may lead to an increase in the frequency of adverse reactions. Therapy should be continued for 24-48 hours after the disappearance of symptoms of the disease and fever.
In case of streptococcal infections, the use of the drug should be continued for 10 days to prevent the development of rheumatic fever or glomerulonephritis.
Children. For children over 12 years of age with a body weight of up to 45 kg, the recommended dose is 4.4 mg/kg of body weight (on the 1st day of treatment, the recommended dose should be administered in one or two doses), on the following days the dose is 2.2 mg/kg of body weight (in one or two doses); in more severe infectious diseases, up to 4.4 mg/kg can be prescribed.
Children weighing 45 kg are given the usual adult dose.
Use of the drug for the treatment of certain infections
Acne: The recommended dose is 50 mg/day with food (including liquid) for 6-12 weeks.
Sexually transmitted diseases: for the treatment of diseases such as uncomplicated gonococcal infections (exception – anorectal infections in men), uncomplicated urethral and endocervical infections and rectal infections caused by Chlamydia trachomatis, non-gonococcal urethritis caused by Ureaplasma urealyticum, the recommended dose is 100 mg 2 times a day for 7 days.
For the treatment of acute epididymo-orchitis caused by Chlamydia trachomatis or Neisseria gonorrhea, the drug should be used at 100 mg 2 times a day for 10 days.
For the treatment of primary and secondary syphilis, the recommended dose for patients without confirmed pregnancy and with an allergy to penicillins is 200 mg orally 2 times a day for 2 weeks (as an alternative to penicillin therapy).
Epidemic relapsing typhus, tick-borne relapsing typhus: the recommended dose is 100-200 mg once, depending on the severity of the disease.
Tropical malaria resistant to chloroquine: the recommended dose is 200 mg/day for at least 7 days due to the potentially severe course of the infectious disease.
A fast-acting schizonticide (e.g. quinine) should always be used as an adjunct to doxycycline, the dose of which varies depending on the case, due to the potentially severe course of the infectious disease.
Malaria prophylaxis: The recommended dose for adults is 100 mg/day. For children over 12 years of age, the recommended dose is 2 mg/kg/day up to a total dose of 100 mg/day. Prophylaxis can be started 1-2 days before travel to a malaria-endemic area. Prophylaxis should be continued daily during travel to a malaria-endemic area and for 4 weeks after leaving the malaria-endemic area. Current malaria treatment guidelines should also be followed.
Prevention of Japanese river fever: the recommended dose is 200 mg once.
Prevention of traveler’s diarrhea in adults: the recommended dose of the drug is 200 mg on the 1st day of travel (used as a single dose of 200 or 100 mg every 12 hours) and 100 mg/day during the following days of travel. There is no information on the use of the drug for prevention purposes for more than 21 days.
Leptospirosis prevention: The recommended dose is 200 mg once a week during the entire stay in the leptospirosis-prone region and 200 mg at the end of the trip. There is no information on the use of the drug for more than 21 days for the purpose of prevention.
Use in the elderly: The drug can be used in normal doses without special precautions. There is no need for dose adjustment in renal impairment. Doxycycline may be the drug of choice for the elderly, as its use is less associated with the development of esophageal irritation and ulcers.
Use of the drug in patients with impaired renal function: use of the drug in recommended doses does not lead to accumulation of the antibiotic in this category of patients (see Features of use).
Use of the drug in patients with impaired liver function: see Features of use.
Contraindication
- Hypersensitivity to doxycycline, to any of the excipients of the drug or to other tetracyclines;
- severe liver dysfunction;
- pregnancy and breastfeeding;
- Patients who have allergic reactions to soy or peanuts should not use this drug.
Side effects
The following adverse reactions have been reported in patients receiving tetracyclines, including doxycycline.
Infections and infestations: superinfection, which can cause candidiasis, glossitis, stomatitis, enterocolitis, pseudomembranous colitis (with increased growth of Clostridium difficile), acute inflammation of the external genitalia and vagina in women (vulvovaginitis), proctitis, itching in the anal area (anal pruritus).
From the hematopoietic and lymphatic system: blood coagulation disorders, decreased prothrombin activity, hemolytic anemia, leukopenia, leukocytosis, thrombocytopenia, anemia, lymphocytopenia, lymphadenopathy, the presence of atypical lymphocytes and toxic granularity in granulocytes, neutropenia, eosinophilia.
On the part of the immune system: photosensitivity reactions, hypersensitivity reactions (including anaphylactic shock, anaphylaxis, angioedema, dyspnea, serum sickness, peripheral edema, tachycardia, urticaria), anaphylactoid reactions, anaphylactoid purpura, pseudoanaphylactic reaction, generalized exanthema, erythema, pruritus, erythema multiforme, reversible local edema of the skin, mucous membranes or joints (angioedema), asthma, local exanthema of the genitals and other areas, malignant exudative erythema, facial edema, tongue edema, laryngeal edema with narrowing of the airways, tachycardia, arterial hypotension, which may progress to shock, severe skin reactions with general reactions (such as exfoliative dermatitis and Lyell’s syndrome), pericarditis, exacerbation of systemic lupus erythematosus. Allergic reactions to soy proteins may occur.
Psychiatric disorders: fear, anxiety, agitation, convulsions (a case of epileptic seizure was reported after oral administration of doxycycline).
Nervous system: headache, paresthesia, malignant intracranial hypertension (Pseudotumor cerebri), which disappears after discontinuation of treatment and is characterized by headache, nausea, vomiting, possible visual disturbances (blurred vision, scotoma, diplopia, temporary loss of vision) due to edema of the optic nerve, flushing, reversible partial loss of olfactory and gustatory sensitivity, prolonged loss of vision. Bulging fontanelle in newborns and benign intracranial hypertension in children and adults have been reported with the use of tetracycline in full therapeutic doses.
From the side of the organs of hearing and vestibular apparatus: ringing in the ears, tinnitus, vertigo.
Cardiovascular system: tachycardia.
Gastrointestinal: nausea, diarrhea, dyspepsia, heartburn, abdominal pain, discoloration of tooth enamel, vomiting, dysphagia, esophagitis, pancreatitis, villous tongue, blackening of the tongue, enamel hypoplasia, bloating, flatulence, steatorrhea, stomatitis, pharyngitis, dysphonia, difficulty swallowing, pseudomembranous enterocolitis, anorexia, irreversible discoloration of teeth and enamel damage. If a doxycycline monohydrate tablet is swallowed incorrectly (for example, in a supine position), esophageal ulcers may occur, as the tablets adhere to the esophageal mucosa and potent acids may be released locally by hydrolysis. Common symptoms of esophagitis and esophageal ulcers include odynophagia, chest pain, and dysphagia.
From the hepatobiliary system: hepatitis, jaundice, liver failure, hepatotoxicity with a temporary increase in liver function tests.
Skin and subcutaneous tissue disorders: exfoliative dermatitis, erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis, rash, including maculopapular and erythematous rashes, skin photosensitivity (redness, swelling, blistering and skin discoloration), photoonycholysis, allergic reaction may involve nails (nail detachment and discoloration), drug hypersensitivity syndrome with eosinophilia and systemic symptoms (DRESS).
Musculoskeletal and connective tissue disorders: myalgia, arthralgia.
Renal and urinary disorders: hematuria, interstitial nephritis, acute renal failure, anuria, increased blood urea. Metabolized tetracyclines have caused a syndrome similar to Fanconi syndrome, including albuminuria, aminoaciduria, hypophosphatemia, hypokalemia, and renal tubular acidosis.
Tetracyclines can cause nephrotoxicity or worsen pre-existing kidney damage (characterized by increased plasma creatinine and urea levels).
Other disorders: with prolonged use of tetracycline, microscopic staining of the thyroid tissue in a brown-black color was noted. However, no pathology of the thyroid gland was detected.
Special instructions
Photosensitivity. Cases of photosensitivity with clinical manifestations of a pronounced sunburn reaction may occur in patients using tetracycline drugs after exposure to direct sunlight or ultraviolet radiation. Patients likely to be exposed to direct sunlight or ultraviolet radiation should be informed about the development of these reactions when using tetracycline drugs with a recommendation to discontinue treatment at the first sign of erythema.
Patients with hepatic impairment. Doxycycline should be used with caution in patients with hepatic impairment and in those receiving concomitant potentially hepatotoxic drugs. Liver function tests have been reported rarely with both oral and parenteral administration of tetracyclines, including doxycycline. In the case of prolonged use and in cases of hepatic impairment, doxycycline plasma levels should be monitored and the dose adjusted if necessary.
Patients with renal impairment. In patients with normal renal function, the renal excretion of doxycycline is approximately 40% per 72 hours. This figure may decrease to 1-5% per 72 hours in patients with severe renal insufficiency (creatinine clearance 10 ml/min). The results of the study indicate that there is no significant difference in T ½ of doxycycline from plasma in patients with normal renal function and in severe renal insufficiency. Hemodialysis does not affect the T ½ of doxycycline from plasma.
The anti-anabolic effect of tetracyclines may increase blood urea levels. Studies have shown that this anti-anabolic effect does not occur when doxycycline is used in patients with severe renal failure.
Tetracyclines can cause nephrotoxicity or worsen existing kidney damage (characterized by increased levels of creatinine and urea in the blood plasma).
Increased growth of microflora. The use of antibiotics in some cases can lead to increased growth of non-susceptible microorganisms, including Candida.
Due to the increasing resistance of many strains of groups of microorganisms, after isolation of the pathogen, it is necessary to check its sensitivity to the drug. In the event of the development of superinfection caused by resistant microorganisms, the antibiotic should be canceled and appropriate therapy should be prescribed.
Pseudomembranous colitis has been reported with nearly all classes of antibacterial agents, including doxycycline. Pseudomembranous colitis has been reported in patients receiving antibacterial agents, including doxycycline. The severity of the disease has ranged from mild to life-threatening. It is important to consider the possibility of this diagnosis in patients with diarrhea secondary to antibacterial therapy.
Clostridium difficile-associated diarrhea (CDAD) has been reported with the use of antibacterial agents, including doxycycline, and can range in severity from mild to fatal colitis. Antibacterial agents disrupt the normal intestinal flora and lead to overgrowth of C. difficile.
C. difficile produces toxins A and B, which, in turn, cause the development of CDAD.
Toxin-producing strains of C. difficile can increase morbidity and mortality because such infections are resistant to antibacterial therapy and may require colectomy. It is important to consider this diagnosis in patients with diarrhea secondary to antibacterial therapy. A careful history is necessary, as CDAD has been reported to develop after 2 months of completion of antibacterial therapy.
Doxycycline may cause tooth discoloration and enamel hypoplasia.
Esophagitis: Esophagitis and esophageal ulcers have been reported with tetracyclines, including doxycycline, in tablet and capsule form. Most patients with these complaints took the drug immediately before bedtime or with insufficient fluid intake.
Porphyria: Porphyria has been reported rarely in patients receiving tetracycline.
Venereal diseases. During treatment for venereal diseases, if a concomitant disease of syphilis is suspected, appropriate diagnostics should be carried out, including dark-field microscopy. In such cases, serological studies should be carried out monthly for at least 4 months.
Some patients with spirochetal infections may develop a Jarisch-Herxheimer reaction shortly after initiation of doxycycline therapy. In such cases, patients should be advised that this reaction is a consequence of antibacterial therapy for spirochetal infections and is usually self-limiting.
Infections caused by β-hemolytic streptococcus. In infections caused by group A β-hemolytic streptococci, treatment should be carried out for at least 10 days.
Myasthenia gravis: Due to the possibility of weak neuromuscular blockade, the drug should be used with caution in patients with myasthenia gravis.
Systemic lupus erythematosus. The use of tetracycline may lead to exacerbation of systemic lupus erythematosus.
Methoxyflurane: Methoxyflurane should be used with caution in combination with tetracyclines.
Benign intracranial hypertension: Bulging fontanelle in neonates and benign intracranial hypertension in adolescents and adults have been reported with full therapeutic doses. The clinical manifestations of these disorders resolved rapidly after discontinuation of doxycycline.
Use during pregnancy or breastfeeding. The drug is contraindicated for use during pregnancy. Doxycycline crosses the placental barrier. The risks associated with the use of tetracyclines during pregnancy are mainly due to their effects on tooth enamel and skeletal bone development.
Tetracycline use during the period of tooth development (pregnancy) may cause permanent discoloration of the teeth (yellow-brown-gray). This adverse reaction is more common with long-term use, but is also possible with repeated short courses of treatment. Enamel hypoplasia has also been reported.
Tetracyclines penetrate into breast milk, so the use of the drug is contraindicated during breastfeeding.
Children. The drug is contraindicated in children under 12 years of age.
Like other tetracyclines, doxycycline forms stable calcium complexes in any bone-forming tissue. Decreased fibular growth has been reported in premature infants given tetracycline orally at a dose of 25 mg/kg every 6 hours. This adverse reaction is reversible upon discontinuation of the drug.
Tetracycline use during the period of tooth development (in children under 12 years of age) may cause permanent tooth discoloration (yellow-brown-gray). This adverse reaction is more common with long-term use, but can also occur with repeated short courses of treatment. Enamel hypoplasia has also been reported.
Ability to influence the speed of reaction when driving vehicles or operating other mechanisms. The effect of doxycycline on the ability to drive vehicles or operate other mechanisms has not been studied. If such undesirable reactions as arterial hypotension, tinnitus, blurred vision, scotoma, diplopia or long-term loss of vision occur, you should refrain from driving vehicles or operating other mechanisms.
Interactions
The absorption of doxycycline in the gastrointestinal tract may be reduced by the simultaneous use of antacids containing aluminum, calcium, magnesium, or other drugs and products containing these cations (for example, milk, dairy products and fruit juices containing calcium), with oral administration of zinc, iron or bismuth salt preparations, activated charcoal or cholestyramine. The use of doxycycline together with such drugs should be maximally spaced out in time.
Bacteriostatic drugs may interfere with the bactericidal effect of penicillin, so it is recommended to avoid the simultaneous use of doxycycline with penicillin.
There have been reports of prolongation of prothrombin time in patients taking anticoagulants (e.g. warfarin, phenprocoumon) and doxycycline. Tetracyclines reduce prothrombin activity in blood plasma, therefore, with such combined use, blood coagulation parameters should be constantly monitored and, if necessary, the doses of anticoagulants should be reduced.
Rifampicin, an inducing agent of the barbiturate class, as well as anticonvulsant drugs such as carbamazepine, diphenylhydantoin and primidone, can accelerate the metabolism of doxycycline in the liver by inducing liver enzymes, as a result of which it will be impossible to achieve therapeutically effective concentrations of doxycycline in the blood when taken in a standard dose.
Alcohol may reduce the T½ of doxycycline.
The drug may enhance the hypoglycemic effect of sulfonylurea derivatives. With such combined use, blood glucose levels should be constantly monitored and, if necessary, the doses of these drugs should be reduced.
Doxycycline may increase the plasma concentration of cyclosporine A, increasing its toxic effect. Careful monitoring is required when these drugs are used simultaneously.
Concomitant use with methotrexate may lead to increased toxicity of the latter.
The simultaneous use of doxycycline with β-lactam antibiotics (penicillins, cephalosporins) should be avoided, as this may lead to a decrease in their antibacterial effectiveness.
Concomitant use of methoxyflurane or other potentially nephrotoxic agents and doxycycline may cause potentially fatal nephrotoxic adverse reactions.
When treating acne with isotretinoin, it is necessary to take a break after doxycycline therapy, as both drugs can in some cases lead to a reversible increase in intracranial pressure.
Concomitant use of theophylline and doxycycline may increase the risk of gastrointestinal side effects.
Doxycycline can lead to both an increase and a decrease in plasma lithium levels when used simultaneously with lithium preparations.
In rare cases, the effectiveness of oral hormonal contraceptives may be reduced. Several cases of pregnancy and breakthrough bleeding have been reported with the concomitant use of doxycycline and oral contraceptives. Therefore, the use of non-hormonal contraceptives is recommended.
Interaction with laboratory tests: the use of tetracycline may lead to inaccurate laboratory test results when determining the level of glucose, protein, urobilinogen and catecholamines in the urine.
Tetracycline can inhibit the breakdown of ergot alkaloids in the liver (individual cases of ergotism are possible).
Because doxycycline contains magnesium, it may enhance the effects of tubocurarine, succinylcholine, and other muscle relaxants.
Overdose
Acute overdose of antibiotics is rare. Overdose can manifest itself in damage to the kidney parenchyma and the development of pancreatitis.
In case of overdose, the drug should be discontinued, gastric lavage and supportive therapy should be performed.
In case of overdose of doxycycline taken orally, it is possible to use antacids, magnesium or calcium salts to bind the unabsorbed part of the drug, since tetracycline can form chelate complexes with these substances that are not absorbed. Dialysis has almost no effect on T ½ from blood plasma, so it is ineffective in overdose.
Storage conditions
At a temperature not exceeding 30°C.
Be the first to review “Doxycycline Teva tablets 100mg blister 10 pcs” Cancel reply
You may also like








Reviews
There are no reviews yet.