No products in the cart.
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
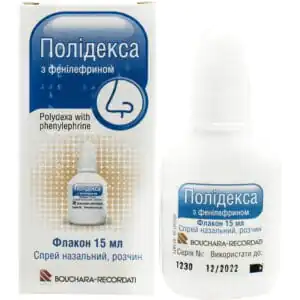
Polydexa with phenylephrine spray nasal solution bottle 15 ml
$31.32
Free Worldwide Shipping
to: Australia, Canada, Czechia, Denmark, Estonia, Ireland, Israel, Italy, Japan, Lithuania, Mexico, Netherlands, Poland, Slovakia, South Korea, Switzerland, United Kingdom, United States and more
In stock
Polydex nasal spray relieves inflammation, swelling, and infection in the nasal cavity using dexamethasone, neomycin, polymyxin B, and phenylephrine.
Categories: Cold and flu
Brand: Bouchara-Recordati
Payment
PayPal, Debit or Credit card, Google Pay, Apple Pay
Pharmacological properties
Pharmacodynamic parameters. The combination of active pharmaceutical ingredients of the drug Polydex determines its anti-inflammatory and antimicrobial effects against a wide range of gram-positive and gram-negative pathogens of infectious-inflammatory diseases of the nasal cavity and paranasal sinuses; helps reduce swelling of the mucous membrane and facilitate nasal breathing.
Synthetic GCS dexamethasone penetrates the cell membrane and binds with high affinity to specific cytoplasmic glucocorticoid receptors. This complex binds to DNA elements, which ultimately leads to modification of transcription and, consequently, protein synthesis to achieve inhibition of leukocyte infiltration at the site of inflammation, interference with the function of inflammatory mediators, suppression of humoral immune reactions and reduction of edema. The anti-inflammatory effect of dexamethasone involves phospholipase A 2- inhibiting proteins lipocortins, which control the biosynthesis of such potent inflammatory mediators as prostaglandins and leukotrienes.
Neomycin is an aminoglycoside antibiotic isolated from cultures of Streptomyces fradiae. Its mechanism of action involves inhibition of protein synthesis by binding to the 30S subunit of the bacterial ribosome, resulting in the bacterium being unable to synthesize proteins vital for its growth. Neomycin may also inhibit bacterial DNA polymerase.
Neomycin is active against B. catarrhalis and Haemophilus influenzae, Acinetobacter, Corynebacterium, Listeria pathogens, Methicillin-resistant Staphylococcus, bacteria of the genera Citrobacter, Enterobacter, Campylobacter, Escherichia coli, Klebsiella, Morganella, Proteus, Providencia, Serratia, Salmonella, Shigella, Yersinia, and Pasteurella.
Neomycin-resistant enterococci, streptococci, methicillin-resistant staphylococci, chlamydia, mycoplasma, rickettsia, P. stuartii, Pseudomonas aeruginosa, A. denitrificans, Burkholderia, N. asteroides, flavobacteria, S. maltophilia, resistant anaerobic microorganisms.
Polymyxin B, a lipopeptide antibiotic isolated from Bacillus polymyxa, has bactericidal activity against Gram-negative bacteria. The α- and γ-diaminobutyric acids of the positively charged polymyxin B form electrostatic interactions with the phosphate groups of the negatively charged lipid A on the outer membrane of Gram-negative bacteria. Calcium and magnesium ions are displaced from the phosphates of membrane lipids, destabilizing the lipopolysaccharide, which increases membrane permeability and, as a result, leads to cell destruction. In addition, polymyxin B inhibits the type II NADH-quinone oxidoreductase on the inner bacterial membrane, which is necessary for cellular respiration, and can also bind and neutralize lipopolysaccharide released during bacterial lysis, preventing the endotoxin response.
Polymyxin B is sensitive to Acinetobacteria, Aeromonas, Alcaligenes spp. microorganisms of the genus Citrobacter, Enterobacteriaceae, Escherichia coli, Klebsiella, Moraxella, Pseudomonas aeruginosa, Salmonella, Shigella, S. maltophilia.
Resistance to polymyxin B is demonstrated by Brucella, Burkholderia (B. cepacia, B. pseudomallei), mycobacteria, B. catarrhalis, campylobacter, legionella, morganella, protea, neisseria, serratia, providencia, C. meningosepticum, Vibrio cholerae biovar El Tor, spherical and rod-shaped spore-forming bacteria.
Phenylephrine is a sympathomimetic amine that acts primarily on α 1 adrenergic receptors of the smooth muscles of the peripheral vascular wall. The vasoconstrictor effect of phenylephrine on the mucous membrane of the nasal cavity and paranasal sinuses leads to a decrease in its swelling and an improvement in the drainage of the paranasal sinuses.
Pharmacokinetic parameters. Due to the insignificant absorption into the systemic bloodstream, the pharmacokinetic parameters of the drug have not been studied.
Indication of Polydexa
Pathology of the nasal cavity of an infectious-inflammatory nature (for example, acute sinusitis, acute rhinitis).
Application
1 injection into each nostril: adults – 3-5 times a day, children aged 15 years – 3 times a day. The duration of the course of therapy is on average 5 days. If there are manifestations of the disease after 10 days of therapy, the use of the drug should be canceled.
The drug is injected in the form of a spray by pressing the bottle. At the time of injection, the bottle should be in a vertical position, the patient’s head should be slightly tilted forward.
Contraindication
- Hypersensitivity to the active pharmaceutical ingredients or other components of the drug; stroke suffered by the patient or the presence of risk factors for its development; angina pectoris (severe or poorly controlled); pronounced mismatch of blood flow in the coronary arteries with the need for oxygen of the heart muscle; assumption that the patient has glaucoma (angle-closure); assumption of the impossibility of complete emptying of the bladder in case of pathology of the prostate gland or urethra; infection with herpes zoster, herpes simplex, varicella virus in the nasal area; seizures (in history); combined use with non-selective MAO inhibitors, any sympathomimetic agents or stimulants α-adrenoblockers; age younger than 15 years.
Side effects
- Allergic reactions and sensitization to antibiotics in the composition of the drug; increased heartbeat, increased heart rate, myocardial infarction; acute attack of glaucoma; vomiting, nausea, xerostomia; stroke, cerebralgia, convulsions; anxiety, agitation, behavioral disorders, hallucinations, insomnia; increased body temperature; urination disorders (in urethroprostatic pathology), urinary retention; hyperhidrosis, itching, rash, urticaria; ag (hypertensive crisis); dryness of the nasal mucosa.
Special instructions
It should not be used orally, and the duration of the course or the dose of the drug should not be increased beyond the recommended amount (the likelihood of a systemic effect as a result of absorption of the active ingredients increases).
The presence of corticosteroids in the drug may affect the symptoms of an allergic reaction.
Dexamethasone in the drug may cause a positive reaction to a doping test.
If necessary, it can be used in combination with antibacterial agents with a systemic effect.
Concomitant use with other vasoconstrictors increases the likelihood of overdose.
Do not use in patients with seizure episodes or concomitantly with drugs that increase the likelihood of a convulsive reaction (local anesthetics, clobutinol, terpene derivatives, drugs containing atropine).
The presence of an active substance with a sympathomimetic effect requires strict adherence to the established course of therapy and dose of the drug, and knowledge of information about contraindications.
Use with caution in patients with hyperthyroidism, coronary artery disease, and hypertension.
Therapy with the drug should be discontinued in the event of nausea or adverse effects from the cardiovascular or nervous system.
Should not be used during pregnancy and breastfeeding.
Interactions of Polydexa
If active substances enter the systemic bloodstream, the following drug interactions are possible.
Phenylephrine should not be combined with non-selective MAO inhibitors (up to 2 weeks after the end of therapy with these agents), any sympathomimetic agents or stimulants, α-adrenoblockers.
It is undesirable to combine phenylephrine with ergot alkaloids, selective and reversible MAO inhibitors, and methylthioninium chloride.
Phenylephrine should be used with caution with halogenated volatile agents for general anesthesia (if possible, discontinue use of the phenylephrine preparation a few days before surgery).
It is undesirable to combine dexamethasone with acetylsalicylic acid.
Dexamethasone should be used with caution with aminoglutemide; praziquantel; drugs that can cause torsade de pointes-type arrhythmia or hypokalemia; digitalis preparations; anticonvulsants, inducers of hepatic metabolic enzymes.
Caution is required when dexamethasone is used concomitantly with NSAIDs, acetylsalicylic acid; antihypertensive drugs; fluoroquinolone antibiotics, some macrolides (erythromycin, telithromycin and clarithromycin); nelfinavir and protease inhibitors boosted with ritonavir; some antifungals (ketoconazole, voriconazole, itraconazole, posaconazole).
Overdose of Polydexa
Systemic effects may develop if the dose of the drug is significantly exceeded or the duration of the course of therapy is increased beyond the recommended one.
Storage conditions
Keep out of reach of children, at a temperature ≤25 °C.
Be the first to review “Polydexa with phenylephrine spray nasal solution bottle 15 ml” Cancel reply
You may also like












Reviews
There are no reviews yet.