No products in the cart.
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more

AEvit (vitamins A+E) soft caps...
$30.09 Original price was: $30.09.$18.99Current price is: $18.99.
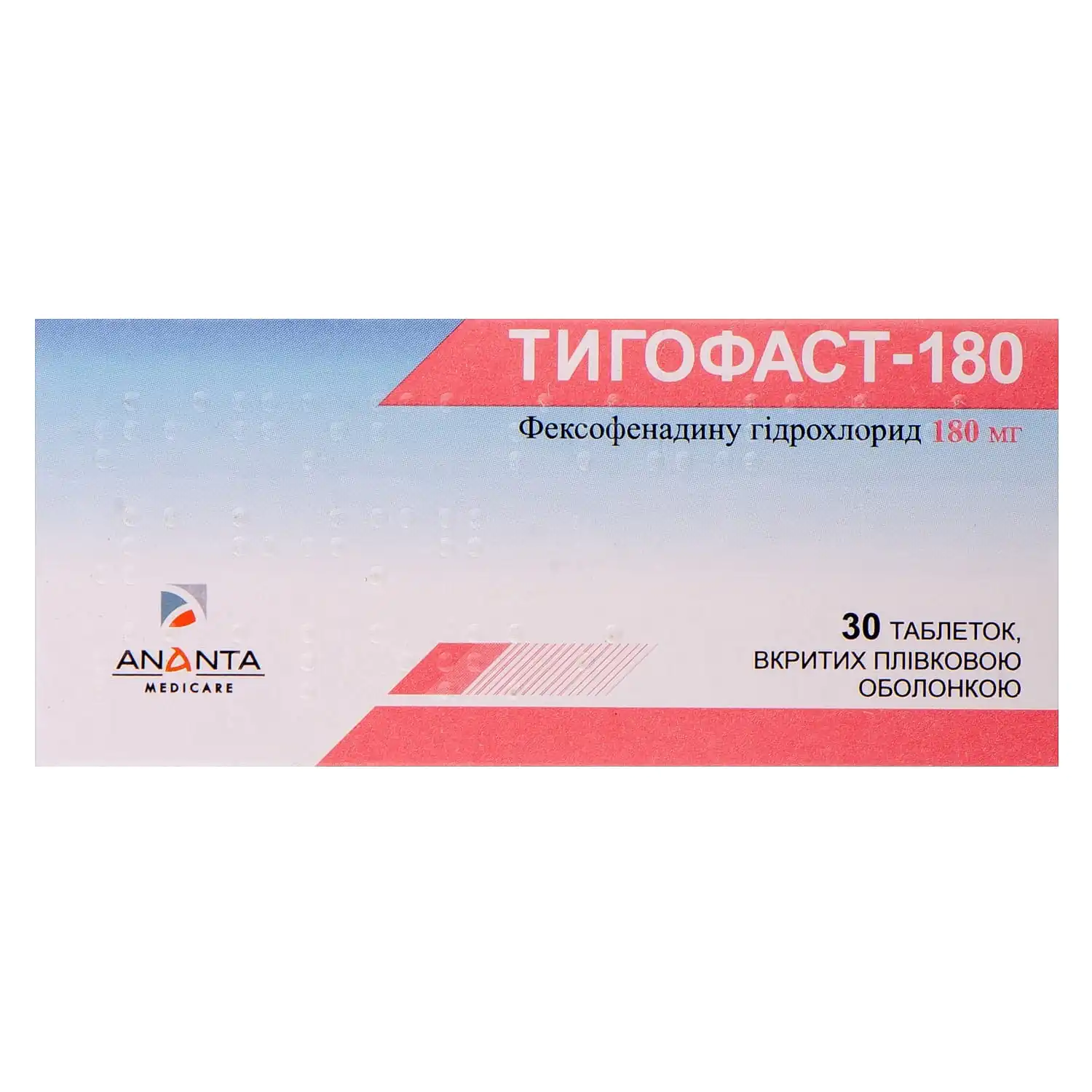
Tigofast 180 film-coated tablets 180 mg 3 blisters of 10 pcs.
$24.43
Free Worldwide Shipping
to: Australia, Canada, Czechia, Denmark, Estonia, Ireland, Israel, Italy, Japan, Lithuania, Mexico, Netherlands, Poland, Slovakia, South Korea, Switzerland, United Kingdom, United States and more
In stock
Tigofast 180 with fexofenadine relieves chronic urticaria symptoms, reducing itching, rash, and allergic reactions without strong sedative effects.
Categories: Allergy, Vitamins and supplements
Brand: Ananta
Payment
PayPal, Debit or Credit card, Google Pay, Apple Pay
Tigofast-180 tablets are used for the symptomatic treatment of chronic idiopathic urticaria in adults and children over 12 years of age.
Composition
The active substance is fexofenadine hydrochloride (one film-coated tablet contains 180 mg of fexofenadine hydrochloride).
Excipients: microcrystalline cellulose, corn starch, sodium starch glycolate (type A), colloidal anhydrous silicon dioxide, Insta Coat Sunset Yellow film coating (ethylcellulose, hydroxypropylmethylcellulose, Sunset Yellow FCF (E 110)), talc.
Contraindication
- hypersensitivity to the components of the drug;
- age up to 12 years.
Method of application
Adults and children over 12 years of age are prescribed the drug for chronic idiopathic urticaria – 180 mg 1 time per day. Take orally before meals, with water. The duration of treatment is determined individually, depending on the severity of the disease.
Application features
Pregnant women
Fexofenadine hydrochloride should not be used during pregnancy unless clearly necessary, when the expected benefit to the mother outweighs the potential risk to the fetus.
Since fexofenadine passes into breast milk, the drug should not be used during breastfeeding.
Children
In this dosage, the drug should not be used in children under 12 years of age.
Drivers
Based on the pharmacodynamic profile and known side effects, it can be concluded that taking this drug does not affect the ability to drive vehicles and perform work requiring concentration of attention. When conducting objective studies, it was found that the drug does not have a significant effect on the functions of the central nervous system (CNS). However, it is recommended to assess the individual response to the drug before starting to drive vehicles or perform work requiring concentration of attention.
Overdose
Most reports of overdose with fexofenadine hydrochloride are not informative enough. For example, dizziness, drowsiness, and dry mouth have been reported.
In case of overdose, the usual measures should be taken to remove unabsorbed active substances. Symptomatic and supportive therapy is recommended. Removal of fexofenadine hydrochloride from the blood by hemodialysis is ineffective.
Side effects
From the nervous system: headache, drowsiness, dizziness.
Gastrointestinal: nausea, diarrhea, epigastric cramps.
General disorders and administration site conditions: feeling of increased fatigue.
Immune system: hypersensitivity reactions, including angioedema, chest tightness, shortness of breath, facial flushing and systemic anaphylactic reactions, dyspnea, flushing.
On the part of the psyche: insomnia, increased irritability and sleep disturbances or unusual dreams (paroniria).
Cardiac: tachycardia, palpitations.
Skin and subcutaneous tissue disorders: rash, exanthema, urticaria, itching.
The medicine contains the dye “Yellow Sunset FCF” (E 110), which may cause allergic reactions.
Storage conditions
Store in the original packaging at a temperature not exceeding 25 °C, out of the reach of children.
Shelf life – 2 years.
Be the first to review “Tigofast 180 film-coated tablets 180 mg 3 blisters of 10 pcs.” Cancel reply
You may also like








Reviews
There are no reviews yet.