No products in the cart.
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more

Chlorophyllipt Farmis Forte sp...
$19.79 Original price was: $19.79.$17.99Current price is: $17.99.
Troxevasin capsules 300 mg 50 pcs.
$25.43
Free Worldwide Shipping
to: Australia, Canada, Czechia, Denmark, Estonia, Ireland, Israel, Italy, Japan, Lithuania, Mexico, Netherlands, Poland, Slovakia, South Korea, Switzerland, United Kingdom, United States and more
In stock
Troxevasin (troxerutin) capsules improve venous tone, reduce swelling, and treat varicose veins and hemorrhoids. Dosage, uses, and precautions.
Categories: Heart and blood vessels, Medicines for children
Brand: Teva
Payment
PayPal, Debit or Credit card, Google Pay, Apple Pay
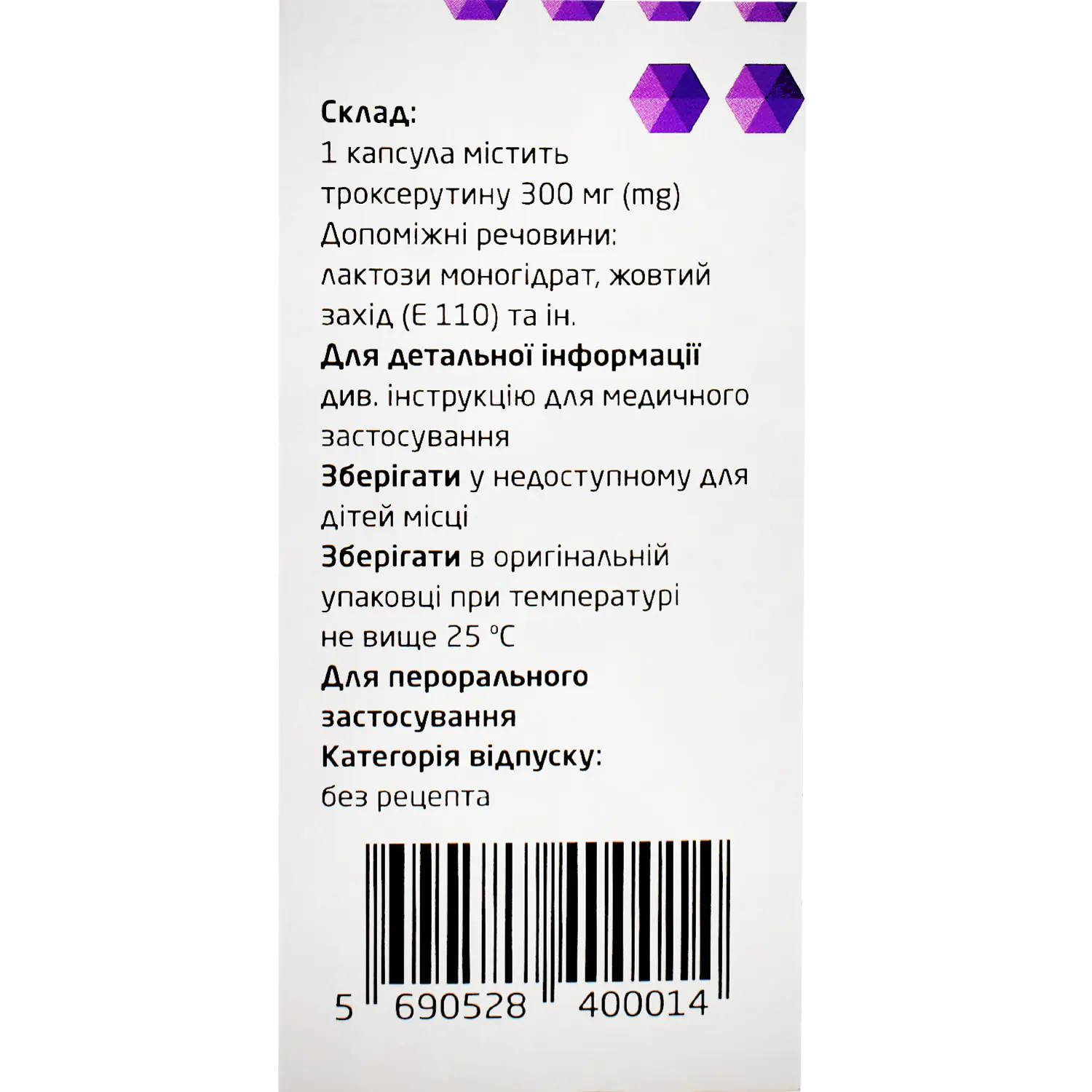
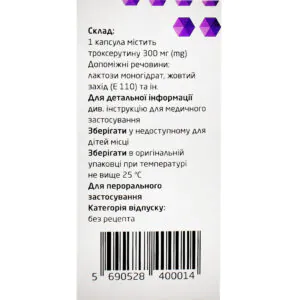
Composition and form of release
Composition
active ingredient: troxerutin;
1 capsule contains troxerutin 300 mg;
excipients: magnesium stearate, lactose monohydrate; quinoline yellow (E 104), sunset yellow (E 110), titanium dioxide (E 171), gelatin.
Release form
Capsules.
Pharmacological properties
Pharmacodynamics. Troxevasin is a mixture of bioflavonoids containing at least 95% troxerutin; capillary tonic and venotonic agent. It exhibits anti-inflammatory and antioxidant effects, inhibits hyaluronidase. Prevents the oxidation of hyaluronic, ascorbic acid and adrenaline, inhibits lipid peroxidation (pol). Troxerutin reduces increased permeability and fragility of capillaries and prevents damage to the basement membrane of endothelial cells by various factors, and in addition, it also has antioxidant, membrane-stabilizing, antihemorrhagic, anti-inflammatory, venotonic effects. Reduces edema, improves trophism, improves microcirculation and microvascular perfusion and other symptoms associated with venous insufficiency or impaired outflow of lymphatic fluid.
Pharmacokinetics. C max in blood plasma is established between 1-9 hours after oral administration. The decrease in plasma concentration is bioexponential. Binding to plasma proteins is within 27-29%, in the largest quantities accumulates in endothelial tissue. Does not penetrate the blood-brain barrier, penetrates the placental barrier in small quantities, in minimal quantities penetrates into breast milk. Metabolized by glucuronidation in the liver. Mainly excreted from the body with bile, in small quantities – with urine.
Indication
Troxevasin capsules are used for symptomatic treatment of the following diseases:
- prevaricose and varicose syndrome, varicose ulcers;
- superficial thrombophlebitis, phlebitis and post-phlebitic conditions;
- chronic venous insufficiency;
- hemorrhoids;
- in complex therapy for diabetic retinopathy;
- swelling and pain due to injuries and varicose veins;
- varicose dermatitis;
- combined treatment for contusions, sprains, dislocations, symptoms of muscle cramps (convulsive contraction of the calf muscles).
Application
Troxevasin capsules are taken orally during meals. The dose and duration of administration of the drug is determined by the doctor depending on the severity of the disease.
The usual dose is 2 capsules per day. Maintenance therapy is 1 capsule per day for 3-4 weeks. This treatment can be combined with the simultaneous use of Troxevasin gel. The effectiveness of treatment with Troxevasin depends on the regularity of administration, the correct dosage and duration of therapy. Clinical experience shows that sometimes the desired effect is observed in doses of 600 mg (2 capsules) per day.
Children: There is no experience with the use of the drug in children.
Contraindication
The drug is contraindicated in patients with hypersensitivity to troxerutin or any of the excipients of the drug; gastric and duodenal ulcers, chronic gastritis (in the acute phase).
Side effects
Adverse reactions may rarely occur during treatment with troxerutin.
On the part of the immune system: skin allergic reactions – rash, itching, urticaria; very rarely – anaphylactic shock, anaphylactoid reactions, hypersensitivity reactions.
From the nervous system: very rarely – dizziness, headache and sleep disturbances.
Vascular disorders: very rarely – ecchymosis.
On the part of the digestive system: rarely possible stomach pain, discomfort in the stomach area, dyspepsia, including nausea, vomiting, flatulence, diarrhea.
General disorders: very rarely – increased fatigue.
Due to the presence of a dye (E110), treatment with the drug may cause an allergic reaction, including asthma.
Special instructions
The drug is ineffective for edema of the lower extremities caused by diseases of the liver, kidneys, or cardiovascular system.
The drug is prescribed with caution to patients with severe liver and gallbladder diseases.
Since the drug can irritate the gastric mucosa, it should be taken with meals.
The drug contains lactose, so it should not be used in patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency or glucose-galactose malabsorption.
The risk of allergy is higher in patients with hypersensitivity to acetylsalicylic acid.
Use during pregnancy or breastfeeding
Pregnancy. There is no data on the negative effects of troxerutin on pregnant and breastfeeding women, but despite this, its use in the first trimester of pregnancy is not recommended.
Breastfeeding. The drug penetrates into breast milk in minimal quantities, but there is no evidence that they caused clinically significant reactions in newborns. The decision on the possibility of using the drug in this category of women is made by the doctor depending on the advantage of the benefit of treatment for the mother over the potential risk to the fetus or infant.
The ability to influence the reaction speed when driving or working with other mechanisms
Troxevasin does not affect the ability to drive vehicles and operate machinery.
Interactions
The drug enhances the effect of ascorbic acid on strengthening the structure and reducing the permeability of the vascular wall.
Overdose
Symptoms: manifestations of adverse reactions may increase.
Treatment. Stop taking the drug. It is recommended to induce vomiting or perform gastric lavage and carry out symptomatic treatment. If necessary, peritoneal dialysis can be prescribed.
Storage conditions
At a temperature not exceeding 25 °C in the original packaging.
Information about the medicinal product for healthcare professionals.
Be the first to review “Troxevasin capsules 300 mg 50 pcs.” Cancel reply
You may also like









Reviews
There are no reviews yet.