No products in the cart.
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
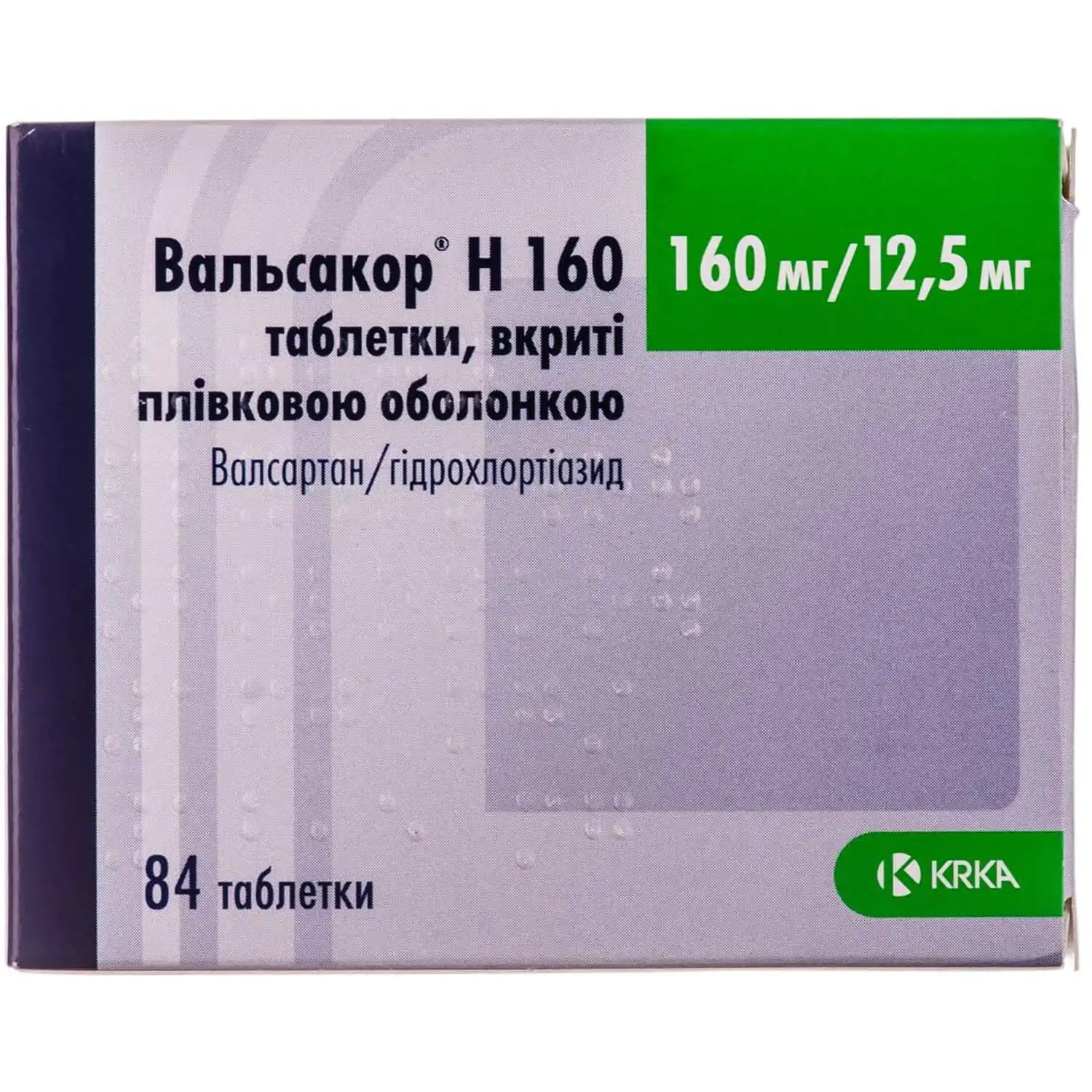
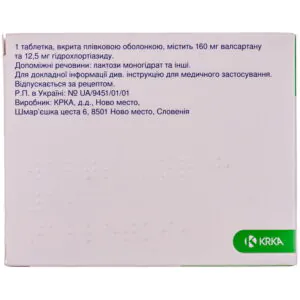
Valsacor H film-coated tablets 160 mg/12.5 mg 84 pieces
$52.14
Free Worldwide Shipping
to: Australia, Canada, Czechia, Denmark, Estonia, Ireland, Israel, Italy, Japan, Lithuania, Mexico, Netherlands, Poland, Slovakia, South Korea, Switzerland, United Kingdom, United States and more
In stock
Valsacor H tablets help control high blood pressure, combining valsartan and hydrochlorothiazide for effective and sustained hypertension treatment.
Categories: Heart and blood vessels
Brand: KRKA
Payment
PayPal, Debit or Credit card, Google Pay, Apple Pay
Pharmacological properties
Pharmacodynamics. valsartan / hydrochlorothiazide. The positive effect of valsartan in combination with hydrochlorothiazide on cardiovascular mortality and morbidity is still unknown.
Epidemiological studies have shown that long-term treatment with hydrochlorothiazide reduces the risk of cardiovascular mortality and morbidity.
Valsartan. Valsartan is a specific antagonist of the angiotensin II (Ang II) receptor. It selectively affects the AT 1 receptor subtype, which is responsible for the known action of angiotensin II. When the concentration of angiotensin II in the blood plasma increases after blockade of the AT 1 receptor due to valsartan administration, the unblocked AT 2 receptor can be stimulated, which balances the effect of the AT 1 receptor . Valsartan does not exhibit any partial agonist action on AT 1 receptors and is characterized by a much greater (about 20,000-fold) affinity for AT 1 receptors than for AT 2 receptors . It is not known whether valsartan binds to and blocks other hormone receptors or ion channels important in cardiovascular regulation.
Valsartan does not inhibit ACE, also known as kininase II, which converts angiotensin I to angiotensin II and leads to the breakdown of bradykinin. Since there is no effect on ACE and there is no potentiation of bradykinin or substance P, angiotensin II antagonists are unlikely to cause cough. When valsartan is compared with an ACE inhibitor, the incidence of dry cough is significantly lower in patients receiving valsartan than in patients receiving ACE inhibitors.
The use of valsartan in patients with hypertension leads to a decrease in blood pressure without affecting the pulse rate. In most patients, after administration of a single oral dose, the onset of the hypotensive effect occurs within 2 hours, and the peak reduction in blood pressure is achieved within 4-6 hours. The hypotensive effect lasts for 24 hours after administration. With repeated administration, the maximum reduction in blood pressure, regardless of the dose, is usually achieved within 2-4 weeks and is maintained with prolonged use. In combination with hydrochlorothiazide, a significant additional reduction in blood pressure is achieved.
Abrupt discontinuation of valsartan does not cause rebound hypertension or other adverse events.
Hydrochlorothiazide. The site of action of thiazide diuretics is mainly the distal convoluted renal tubule. There is a high-affinity receptor in the renal cortex that serves as the primary binding site for thiazide diuretics and inhibition of NaCl transport in the distal convoluted renal tubule. The mechanism of action of thiazides is through inhibition of the Na + Cl – symporter, probably due to competition for the Cl – site , which affects the mechanisms of electrolyte reabsorption: a direct increase in sodium levels and chloride excretion to approximately the same extent and an indirect effect of this diuretic, which reduces the level in the blood plasma with a subsequent increase in plasma renin activity, aldosterone secretion and a decrease in urinary potassium, as well as a decrease in plasma potassium. The relationship between renin and aldosterone is mediated by angiotensin II, therefore, with simultaneous use of valsartan, the decrease in potassium in the blood plasma is less pronounced than with monotherapy with hydrochlorothiazide.
Pharmacokinetics. Valsartan / hydrochlorothiazide. The systemic availability of hydrochlorothiazide is reduced by approximately 30% when co-administered with valsartan. The kinetics of valsartan were not significantly affected by co-administration with hydrochlorothiazide. This apparent interaction does not affect the combined use of valsartan and hydrochlorothiazide, since the clear hypotensive effect of the combination exceeds that achieved with monotherapy with each of the active substances or placebo.
Valsartan. Absorption. After oral administration of valsartan, peak plasma concentrations are reached within 2-4 hours. The mean absolute bioavailability is 23%. Food intake reduces the exposure (measured as AUC) of valsartan by approximately 40% and the plasma Cmax by approximately 50%, although starting from approximately 8 hours after administration, plasma valsartan concentrations are similar in fed and fasted patients. This decrease in AUC, however, is not accompanied by a clinically significant decrease in therapeutic effect, and valsartan can be taken without regard to food intake.
Distribution. The steady-state volume of distribution of valsartan after intravenous administration is 17 liters, indicating that valsartan is not actively distributed into tissues. Valsartan is actively bound to plasma proteins (94-97%), particularly plasma albumin.
Biotransformation: Valsartan is biotransformed to a negligible extent, since only 20% of the dose is recovered as metabolites. The hydroxyl metabolite is found in plasma in small concentrations (10% of the AUC for valsartan). This metabolite is pharmacologically inactive.
Elimination. Valsartan exhibits multiexponential decay kinetics (T ½ α after 1 h and T ½ ß after approximately 9 h). Valsartan is excreted mainly in feces (about 83% of the dose) and urine (about 13% of the dose) unchanged. After administration, the clearance of valsartan from blood plasma is ≈2 l / h, and renal clearance is 0.62 l / h (about 30% of the total clearance). T ½ valsartan is 6 h.
Hydrochlorothiazide. Absorption. Hydrochlorothiazide is rapidly absorbed after oral administration (Tmax ≈2 h), with similar absorption characteristics for the suspension and tablet formulations. The bioavailability of hydrochlorothiazide is 60-80% after oral administration. Co-administration with food has been reported to result in both increased and decreased systemic bioavailability of hydrochlorothiazide compared to fasting conditions. The frequency of these effects is low and of minimal clinical significance. The increase in mean AUC is linear and dose-proportional over the therapeutic dose range. There is no change in the kinetics of hydrochlorothiazide with repeated dosing, and accumulation is minimal with once-daily dosing.
Distribution. The kinetics of distribution and elimination are generally described by a biexponential descending curve. The volume of distribution is 4-8 l/kg. Hydrochlorothiazide in the general circulation binds to plasma proteins (40-70%), mainly to plasma albumin.
Hydrochlorothiazide also accumulates in erythrocytes, approximately 1.8 times higher than the level in blood plasma.
Excretion: More than 95% of the absorbed dose of hydrochlorothiazide is excreted unchanged in the urine. Renal excretion consists of passive filtration and active secretion in the renal tubules. The final T½ is 6-15 hours.
Special patient groups
Elderly: Slightly higher systemic exposure to valsartan has been observed in some elderly subjects compared to younger subjects; however, this has not been shown to be of clinical significance.
Limited data indicate that the systemic clearance of hydrochlorothiazide is reduced in both healthy elderly subjects and elderly subjects with hypertension compared to healthy young volunteers.
Renal impairment: At the recommended dose of valsartan/hydrochlorothiazide, no dose adjustment is required for patients with creatinine clearance 30-70 ml/min.
There are no data for valsartan/hydrochlorothiazide in patients with severe renal impairment (creatinine clearance 30 ml/min) and patients undergoing dialysis. Valsartan is highly bound to plasma proteins and is not dialysable, while hydrochlorothiazide is cleared by dialysis.
Renal clearance of hydrochlorothiazide consists of passive filtration and active secretion in the renal tubules. As expected for a substance that is almost entirely excreted by the kidneys, renal function has a marked effect on the kinetics of hydrochlorothiazide.
Hepatic impairment: There are no data on the use of valsartan in patients with severe hepatic impairment. Liver disease does not significantly affect the pharmacokinetics of hydrochlorothiazide.
Indication
Ag in patients whose blood pressure is not regulated as a result of monotherapy.
Application
The recommended dose is 1 tablet of Valsacor N80 (80 mg / 12.5 mg) per day. In case of insufficient reduction of HBP after 3-4 weeks after the start of treatment, it is recommended to consider increasing the dose to 1 tablet of Valsacor h160 (160 mg / 12.5 mg) 1 time per day. Valsacor hd160 (160 mg / 25 mg) should be prescribed to patients who do not achieve sufficient reduction of HBP when using Valsacor h160.
In case of insufficient blood pressure reduction when taking Valsacor HD160, treatment with valsartan / hydrochlorothiazide tablets 320 mg / 12.5 mg (Valsacor H320) should be started. Valsartan / hydrochlorothiazide tablets 320 mg / 25 mg (Valsacor HD320) should be used in patients whose blood pressure control is insufficient when taking valsartan / hydrochlorothiazide tablets 320 mg / 12.5 mg.
The maximum daily dose is 320 mg / 25 mg.
The antihypertensive effect is mainly observed in the first 2 weeks. In most patients, the maximum effect is observed within 4 weeks. However, some patients may require 4-8 weeks of treatment. This should be taken into account when selecting the dose.
Valsacor H80, Valsacor H160, Valsacor HD160, Valsacor H 320 and Valsacor HD 320 can be taken regardless of meals, with water.
Renal impairment. No dose adjustment is required for patients with mild to moderate renal impairment (glomerular filtration rate (GFR) ≥30 mL/min). Due to the hydrochlorothiazide content, the drug is contraindicated in patients with severe renal impairment (GFR 30 mL/min) and anuria (see Adverse Reactions, Interactions and Pharmacokinetics). Concomitant use with aliskiren is contraindicated in patients with renal impairment (GFR 60 mL/min/1.73 m2 ) (see Adverse Reactions).
Hepatic impairment. In patients with mild to moderate hepatic impairment without cholestasis, the dose of valsartan should not exceed 80 mg (see Precautions). No dose adjustment of hydrochlorothiazide is required in patients with mild to moderate hepatic impairment. Due to the valsartan content, the drug is contraindicated in patients with severe hepatic impairment or biliary cirrhosis and cholestasis (see Side Effects, Precautions and Pharmacokinetics).
Diabetes mellitus. The concomitant use of valsartan and aliskiren is contraindicated in patients with diabetes mellitus (see Adverse Reactions).
Elderly patients: No dosage adjustment is required for elderly patients.
Contraindication
Hypersensitivity to the components of the drug, hypersensitivity to sulfonamide derivatives; severe liver dysfunction, cirrhosis and cholestasis, anuria, severe renal dysfunction (creatinine clearance 30 ml/min); refractory hypokalemia, hyponatremia, hypercalcemia or symptomatic hyperuricemia; simultaneous use of angiotensin receptor antagonists, including valsartan or ACE inhibitors, with aliskiren in patients with diabetes mellitus or renal failure (GFR 60 ml/min/1.73 m2); pregnant women or women planning pregnancy (see use during pregnancy and breastfeeding).
Side effects
The following are listed by organ system adverse reactions that were more common with the combination of valsartan and hydrochlorothiazide than with placebo, and were reported in clinical trials and laboratory observations, as well as individual postmarketing reports. Adverse reactions that occurred with each component alone but were not observed in clinical trials may occur with the combination of valsartan and hydrochlorothiazide.
Adverse reactions are classified according to frequency of occurrence: very common (≥1/10); common (≥1/100, 1/10); uncommon (≥1/1000, 1/100); rare (≥1/10,000, 1/1000); very rare (1/10,000); unknown (cannot be determined from available data).
Within each frequency grouping, adverse reactions are presented in order of decreasing seriousness.
Frequency of adverse reactions when taking valsartan / hydrochlorothiazide
Metabolism and digestion: infrequently – dehydration.
From the nervous system: very often – dizziness, headache; infrequently – paresthesia; unknown – loss of consciousness.
On the part of the organ of vision: infrequently – blurred vision, blurred vision, visual impairment.
On the part of the organ of hearing: infrequently – tinnitus, otitis media.
From the vascular system: infrequently – arterial hypotension.
Respiratory, thoracic and mediastinal disorders: infrequently – cough, nasopharyngitis, upper respiratory tract infection.
not known: non-cardiogenic pulmonary edema, sinusitis, bronchitis.
Gastrointestinal tract: very rarely – diarrhea.
Musculoskeletal and connective tissue disorders: infrequently – muscle pain; very rarely – arthralgia, back pain.
Renal and urinary disorders: unknown – renal dysfunction.
General disorders and administration site conditions: infrequently – fatigue.
Laboratory tests: unknown – increased uric acid in blood plasma, increased bilirubin and creatinine levels in blood plasma, hypokalemia, hyponatremia, increased blood urea nitrogen, neutropenia.
Additional information on individual components: Adverse reactions previously reported with one of the individual components may also occur during treatment with the drug, even if they were not observed during clinical trials or during the post-marketing period.
Frequency of adverse reactions when taking valsartan
From the side of the circulatory and lymphatic system: unknown – decreased hemoglobin, decreased hematocrit, thrombocytopenia.
Immune system disorders: unknown – other hypersensitivity/allergy reactions, including serum sickness.
Metabolism and digestion: unknown – increased serum potassium levels.
On the part of the organ of hearing: infrequently – vertigo.
From the vascular system: unknown – vasculitis.
Gastrointestinal tract: infrequently – abdominal pain.
From the liver and biliary tract: unknown – increased liver function tests.
Skin and subcutaneous tissue disorders: not known – angioedema, rash, itching.
Renal and urinary disorders: unknown – renal failure.
Frequency of adverse reactions when taking hydrochlorothiazide
From the side of the blood and lymphatic system: rarely – thrombocytopenia, sometimes with purpura; very rarely – agranulocytosis, leukopenia, hemolytic anemia, bone marrow suppression.
On the part of the immune system: very rarely – hypersensitivity reactions.
Mental disorders: rarely – depression, sleep disorders.
From the nervous system: rarely – headache, dizziness, paresthesia, visual disturbances.
From the cardiovascular system: rarely – cardiac arrhythmias.
From the vascular system: often – postural hypotension.
From the respiratory, thoracic and mediastinal system: very rarely – respiratory arrest, including pneumonitis and pulmonary edema.
From the digestive system: often – loss of appetite, mild nausea and vomiting; rarely – constipation, discomfort in the digestive tract; very rarely – pancreatitis.
From the liver and biliary tract: rarely – intrahepatic cholestasis or jaundice.
Skin and subcutaneous tissue disorders: common: urticaria and other forms of rash; rare: photosensitivity; very rare: necrotizing vasculitis and toxic epidermal necrolysis of the skin, skin reactions similar to systemic lupus erythematosus, reactivation of skin manifestations of lupus erythematosus.
From the reproductive system and mammary glands: often – impotence.
Electrolyte and metabolic disorders: Hypokalemia has been reported in patients treated with thiazide diuretics. Thiazide diuretic therapy is frequently associated with hyponatremia and hypochloremic alkalosis. Thiazides increase urinary magnesium excretion, which may result in hypomagnesemia.
If severe side effects occur, the drug should be discontinued.
Special instructions
Renal impairment and kidney transplantation. Patients with a kidney transplant should not take the drug. For people with impaired renal function, no dose adjustment is required if creatinine clearance ≥30 ml/min. Due to the hydrochlorothiazide content, Valsacor h80, Valsacor h160, Valsacor hd160, Valsacor n 320 and Valsacor НD 320 are contraindicated in patients with severe renal insufficiency (creatinine clearance 30 ml/min) and anuria. It is recommended to periodically monitor the level of potassium, creatinine and uric acid in the blood plasma.
Thiazides can provoke azotemia in patients with chronic renal failure.
Renal artery stenosis. Since there is a possibility of an increase in blood urea and creatinine levels in the blood plasma, patients with unilateral or bilateral renal artery stenosis or stenosis of a solitary kidney are not recommended to take the drug.
Primary aldosteronism. The drug is not recommended for use in patients with primary aldosteronism, since the underlying disease affects the renin-angiotensin-aldosterone system (RAAS).
Patients with severe congestive heart failure or other conditions associated with RAAS stimulation. In patients whose renal function may depend primarily on RAAS activity (e.g., patients with severe congestive heart failure), the use of agents acting on the RAAS may cause oliguria and/or progressive azotemia, in rare cases with the development of acute renal failure. The safety of the drug in patients with severe congestive heart failure has not been established. Therefore, it cannot be ruled out that renal failure may develop due to RAAS inhibition with the use of Valsacor H80, Valsacor H160, Valsacor HD160, Valsacor H 320 and Valsacor HD 320. Such patients should not take the drug.
Changes in the balance of electrolytes in the blood plasma. Caution should be exercised when taking potassium supplements; potassium-sparing diuretics; salt substitutes containing potassium; or other drugs that may increase the concentration of potassium in the blood (heparin).
Hypokalemia has been reported during treatment with thiazide diuretics. Regular monitoring of plasma potassium levels is recommended.
Hydrochlorothiazide: Hypokalemia has been reported during treatment with thiazide diuretics, including hydrochlorothiazide. Regular monitoring of plasma potassium levels is recommended.
Treatment with thiazide diuretics, including hydrochlorothiazide, causes hyponatremia and hypochloremic alkalosis. Thiazides, including hydrochlorothiazide, increase urinary magnesium excretion, which may lead to hypomagnesemia. Calcium excretion is decreased by thiazides. This may lead to hypercalcemia.
In patients receiving diuretic therapy, periodic assessment of plasma electrolyte levels should be performed at appropriate frequency.
Plasma electrolyte levels should be checked regularly.
Patients with sodium and/or volume depletion. Patients taking thiazide diuretics should be observed for clinical signs of fluid or electrolyte imbalance. Warning signs of fluid and electrolyte imbalance include dry mouth, thirst, asthenia (fatigue, weakness), drowsiness, lethargy, agitation (dysphoria), muscle cramps or pain, muscle fatigue, hypotension, oliguria, tachycardia, and gastrointestinal disturbances (nausea and vomiting).
In patients with severe sodium and/or volume depletion (e.g., high-dose diuretics), symptomatic hypotension may occur in some cases after initiation of therapy with the drug. Correction of sodium and/or volume depletion should be performed prior to initiation of therapy. In case of hypotension, the patient should be placed in a horizontal position with the lower extremities elevated and parenteral saline should be administered. Treatment may be continued after stabilization of blood pressure.
Aortic and mitral valve stenosis, obstructive hypertrophic cardiomyopathy: Particular caution should be exercised in patients with aortic or mitral valve stenosis or obstructive hypertrophic cardiomyopathy.
Hepatic impairment. In patients with mild or moderate hepatic impairment, the drug should be used with caution, not more than 80 mg of valsartan per day.
Due to the content of valsartan, Valsacor H80, Valsacor H160, Valsacor HD160, Valsacor H 320 and Valsacor HD 320 are contraindicated in patients with severe hepatic insufficiency or biliary cirrhosis and cholestasis. In patients with mild to moderate hepatic impairment without cholestasis, no dose adjustment of hydrochlorothiazide is required. However, the drug should be used with caution. Liver disease has little effect on the pharmacokinetics of hydrochlorothiazide. Thiazides should be used with caution in patients with impaired hepatic function or progressive liver disease, since minor changes in fluid or electrolyte balance may lead to hepatic coma.
Systemic lupus erythematosus: Exacerbation or activation of systemic lupus erythematosus has been reported during treatment with thiazides.
Other metabolic disorders: Thiazides may affect glucose tolerance and increase plasma cholesterol, triglycerides, and uric acid levels. Patients with diabetes may require adjustment of insulin or hypoglycemic drugs.
Thiazides may reduce urinary calcium excretion and cause intermittent and slight increases in plasma calcium levels in the absence of known disorders of calcium metabolism. Marked hypercalcemia may indicate the presence of hyperparathyroidism. Thiazides should be discontinued before parathyroid function tests are performed.
Photosensitivity. The development of photosensitivity has been noted when taking thiazides (see Side effects). In the event of a photosensitivity reaction during treatment with the drug, it is recommended to discontinue therapy. If it is necessary to take the diuretic again, it is recommended to protect vulnerable areas from solar or artificial UV radiation.
Acute angle-closure glaucoma. Hydrochlorothiazide has been associated with idiosyncratic reactions resulting in acute transient myopathy and acute glaucoma. Symptoms include sudden loss of visual acuity or eye pain, usually occurring within hours to 1 week after administration. Untreated acute glaucoma can lead to permanent vision loss.
Hydrochlorothiazide treatment should be discontinued as soon as possible. Urgent medical or surgical attention may be required if intraocular pressure remains uncontrolled. Risk factors for the development of acute glaucoma may include a history of allergy to sulfonamides or penicillin.
General disorders: Caution should be exercised when using the drug in patients with hypersensitivity to other angiotensin II receptor antagonists. In patients with a history of allergy and asthma, there is a high probability of developing allergic reactions.
Angioedema. Angioedema (including laryngeal and glottis swelling leading to airway obstruction and/or swelling of the face, lips, pharynx and/or tongue) has been reported in patients treated with valsartan. Some of these patients had a history of angioedema with other medicinal products, including other angiotensin II receptor antagonists. If angioedema develops, treatment with angiotensin II receptor antagonists should be discontinued immediately. Re-administration is contraindicated.
Dual blockade of the RAAS. Hypertension, syncope, stroke, hyperkalemia and changes in renal function, including acute renal failure, have been observed in susceptible patients, especially with the combination of drugs that affect this system. Due to dual blockade of the RAAS, the concomitant use of aliskiren and angiotensin II receptor antagonists or ACE inhibitors is not recommended.
Hydrochlorothiazide may reduce the level of protein-bound iodine in blood plasma.
Hydrochlorothiazide is capable of increasing the concentration of free bilirubin in blood plasma.
Elderly patients do not require dose adjustment.
Important information about some of the ingredients. Valsacor H80, Valsacor H160, Valsacor HD160, Valsacor H 320 and Valsacor HD 320 contain lactose. Patients with rare hereditary forms of galactose intolerance, the Lapp lactase deficiency or glucose-galactose malabsorption should not take this medicine.
Use during pregnancy and breastfeeding
pregnancy
Valsartan. The use of angiotensin II receptor antagonists is contraindicated during pregnancy. Epidemiological data on the risk of teratogenicity due to the action of ACE inhibitors during the first trimester of pregnancy are inconclusive; however, a small increase in risk cannot be excluded. Unless continued therapy with angiotensin II receptor antagonists is considered essential, patients planning pregnancy should be changed to alternative antihypertensive treatments which have an established safety profile for use in pregnancy. When pregnancy is diagnosed, angiotensin II receptor antagonists should be stopped immediately, and, if appropriate, alternative therapy should be started.
Exposure to angiotensin II receptor antagonists during the second and third trimesters of pregnancy has been shown to induce human fetotoxicity (decreased renal function, oligohydramnios, skull ossification retardation) and neonatal toxicity (renal failure, hypotension, hyperkalemia). Infants whose mothers have taken angiotensin II receptor antagonists should be closely monitored for hypotension.
Hydrochlorothiazide. Experience with the use of hydrochlorothiazide during pregnancy, especially in the third trimester, is limited. Animal data are insufficient. Hydrochlorothiazide crosses the placental barrier. Given the pharmacological mechanism of action of hydrochlorothiazide, its use in the second and third trimesters may lead to impaired fetoplacental circulation and cause fetal and neonatal effects such as jaundice, impaired water-salt balance and thrombocytopenia.
Breastfeeding
Information on the use of valsartan during breastfeeding is not available. Hydrochlorothiazide is excreted in human milk in small amounts. Thiazides in high doses, causing intense diuresis, may suppress milk production. Therefore, the use of valsartan / hydrochlorothiazide during breastfeeding is not recommended. Alternative treatments with an established safety profile during breastfeeding are preferable, especially while nursing a newborn or preterm infant.
Children. The safety and effectiveness of the drug in children have not been established, therefore it is not used in pediatric practice.
The ability to influence the reaction rate when driving vehicles or working with other mechanisms. At the beginning of the use of the drug (the period is determined by the doctor), it is forbidden to drive vehicles and work with other mechanisms. Later, the degree of prohibition is determined by the doctor.
Interactions
Interactions with the combination of valsartan and hydrochlorothiazide
Concomitant use is not recommended.
Lithium: Reversible increases in plasma lithium concentrations and toxicity have been reported with concomitant use of lithium and ACE inhibitors and/or thiazides. There is no experience with the concomitant use of valsartan and lithium; therefore, monitoring of plasma lithium concentrations is recommended.
Concomitant use requiring special precautions
Other antihypertensive drugs. The drug may enhance the hypotensive effect of other antihypertensive drugs (e.g. ACE inhibitors, β-adrenergic blockers, calcium channel blockers).
Pressor amines (e.g., noradrenaline, adrenaline). Significantly reduced response to pressor amines. The clinical significance of this effect is not known for certain and is not sufficient to preclude their use.
NSAIDs, including selective COX-2 inhibitors, acetylsalicylic acid (3 g/day) and non-selective NSAIDs. NSAIDs may reduce the hypotensive effect of both angiotensin II antagonists and hydrochlorothiazide when used concomitantly. In addition, concomitant use of valsartan/hydrochlorothiazide and non-steroidal anti-inflammatory drugs may lead to a decrease in renal function and plasma potassium levels. Therefore, it is recommended to monitor renal function at the beginning of treatment, and to ensure that the patient is adequately hydrated.
Interactions related to valsartan
Dual blockade of the RAAS with angiotensin II receptor antagonists, ACE inhibitors or aliskiren. Caution should be exercised when administering angiotensin II receptor antagonists, including valsartan, with other drugs that block the RAAS, such as ACE inhibitors or aliskiren (see Precautions).
The concomitant use of angiotensin receptor antagonists, including valsartan, or ACE inhibitors with aliskiren is contraindicated in patients with diabetes mellitus or renal impairment (GFR < 60 ml/min/1.73 m2 ) (see Adverse Reactions).
Concomitant use is not recommended.
Potassium-sparing diuretics, potassium supplements, salt substitutes containing potassium, and other substances that may increase potassium levels. Caution should be exercised when concomitantly taking drugs that affect potassium levels. Frequent monitoring of plasma potassium levels is recommended.
lack of interactions
In studies with valsartan, no clinically significant interactions were observed when valsartan was administered with any of the following substances: cimetidine, warfarin, furosemide, digoxin, atenolol, indomethacin, hydrochlorothiazide, amlodipine and glibenclamide. Digoxin and indomethacin may interact with hydrochlorothiazide in the formulations Valsacor H80, Valsacor H160, Valsacor HD160, Valsacor H320 and Valsacor HD320 (see Interactions related to hydrochlorothiazide).
Interactions related to hydrochlorothiazide
Concomitant use requiring special attention
Drugs affecting plasma potassium levels. The hypokalemic effect of hydrochlorothiazide may be enhanced by concomitant use with saluretics, corticosteroids, laxatives, ACTH, amphotericin, carbenoxolone, penicillin G, salicylic acid and its derivatives.
If the above drugs are prescribed in combination with hydrochlorothiazide / valsartan, it is recommended to monitor the level of potassium in the blood plasma (see Special warnings and precautions for use).
Drugs that can cause torsades de pointes tachycardia:
- Class Ia antiarrhythmic drugs (quinidine, hydroquinidine, disopyramide);
- class III antiarrhythmic drugs (amiodarone, sotalol, dofetilide, ibutilide);
- some neuroleptics (e.g. thioridazine, chlorpromazine, levomepromazine, trifluoperazine, cyamemazine, sulpiride, sultopride, amisulpride, tiapride, pimozide, haloperidol, droperidol);
- others (e.g. bepridil, cisapride, diphemanil, erythromycin IV, halofantrine, ketanserin, mizolastine, pentamidine, sparfloxacin, terfenadine, vincamine IV).
Due to the risk of hypokalemia, hydrochlorothiazide should be used with caution simultaneously with drugs that can induce torsades de pointes tachycardia.
Drugs affecting plasma sodium levels. The hyponatremic effect of diuretics may be enhanced by concomitant use of drugs such as antidepressants, antipsychotics, and antiepileptics. Long-term use of these drugs should be undertaken with extreme caution.
Cardiac glycosides. In case of arrhythmia caused by digitalis preparations while taking thiazides, hypokalemia or hypomagnesemia may develop (see Precautions).
Calcium salts and vitamin D. The use of thiazide diuretics, including hydrochlorothiazide, in combination with vitamin D or calcium salts may cause an increase in plasma calcium levels. The simultaneous use of thiazide diuretics with calcium salts may lead to hypercalcemia in patients with a predisposition to hypercalcemia (for example, patients with hyperparathyroidism, malignant tumors or conditions caused by vitamin D deficiency) by increasing tubular calcium reabsorption.
Antidiabetic medicinal products (oral agents and insulin): Thiazides may alter glucose tolerance. Dose adjustment of antidiabetic medicinal products may be required.
Metformin should be used with caution due to the risk of lactic acidosis caused by possible functional renal failure associated with hydrochlorothiazide.
Beta-blockers and diazoxide: Concomitant use of thiazide diuretics, including hydrochlorothiazide, with beta-blockers may increase the risk of hyperglycemia. Thiazides, including hydrochlorothiazide, may potentiate the hyperglycemic effect of diazoxide.
Medicinal products used in the treatment of gout (probenecid, sulfinpyrazone and allopurinol). There is a need for dose adjustment of drugs for the treatment of gout, since hydrochlorothiazide may increase the level of uric acid in the blood plasma. If necessary, the dose of probenecid or sulfinpyrazone should be increased. Concomitant administration of thiazide diuretics may increase the frequency of hypersensitivity reactions to allopurinol.
Anticholinergics (e.g. atropine, biperiden). The bioavailability of thiazide diuretics may be increased by anticholinergics (e.g. atropine, biperiden), in particular due to a decrease in gastric motility and rate of gastric emptying. In turn, prokinetic drugs, e.g. cisapride, are expected to reduce the bioavailability of thiazide diuretics.
Amantadine: Thiazides increase the risk of side effects of amantadine.
Ion exchange resins: Absorption of thiazide diuretics, including hydrochlorothiazide, is impaired by concomitant administration of cholestyramine or colestipol.
Cytotoxic drugs (e.g. cyclophosphamide, methotrexate). Thiazides may reduce the renal excretion of cytotoxic drugs and potentiate their myelosuppressive effect.
Non-depolarizing skeletal muscle relaxants (e.g. tubocurarine). Thiazides potentiate the action of curare derivatives.
Cyclosporine: Concomitant use with cyclosporine may increase the risk of hyperuricemia and the appearance of symptoms resembling gout flares.
Alcohol, barbiturates, or narcotics: Concomitant use of thiazide diuretics with substances that also lower blood pressure (e.g., by decreasing sympathetic nervous system activity or by direct vasodilating action) may potentiate orthostatic hypotension.
Methyldopa: Cases of hemolytic anemia have been reported with concomitant use of hydrochlorothiazide and methyldopa.
Carbamazepine: Hyponatremia may develop in patients receiving hydrochlorothiazide concomitantly with carbamazepine. Such patients should be informed of the possibility of hyponatremia and monitored appropriately.
Iodine preparations: In case of dehydration due to diuretics, the risk of acute renal failure increases, especially with high doses of iodine preparations. Patients should be rehydrated before administration.
Overdose
Symptoms. Overdose of valsartan can lead to severe hypotension, which, in turn, can contribute to depression of consciousness, the development of vascular insufficiency and / or shock. The following signs and symptoms may occur as a result of an overdose of hydrochlorothiazide: nausea, drowsiness, hypovolemia, electrolyte imbalance and, as a result, arrhythmia and muscle spasms. The most characteristic signs and symptoms of overdose are also tachycardia, arterial hypotension, shock, weakness, confusion, dizziness, muscle spasms, paresthesia, exhaustion, disorders of consciousness, vomiting, thirst, polyuria, oliguria, anuria, alkalosis, increased blood urea nitrogen (mainly renal failure).
Treatment. Therapeutic measures depend on how long ago the high dose of the drug was taken and the severity of the symptoms, with the most important being stabilization of hemocirculation.
If the drug was taken recently, vomiting should be induced. If a long time has passed since ingestion, activated charcoal should be administered in sufficient quantities.
In the event of arterial hypotension, the patient should be placed in a horizontal position and immediately administered intravenously with isotonic saline to restore water-salt balance.
Valsartan is not removed by hemodialysis due to its strong binding to plasma proteins, however, clearance of hydrochlorothiazide is achieved by dialysis.
Storage conditions
In the original packaging at a temperature not exceeding 30 °C.
Be the first to review “Valsacor H film-coated tablets 160 mg/12.5 mg 84 pieces” Cancel reply
You may also like








Reviews
There are no reviews yet.