No products in the cart.
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
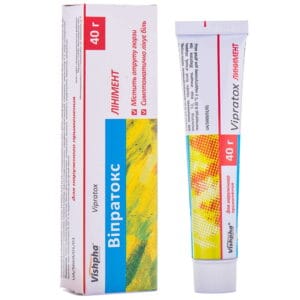
Diclosafe Forte emulsion gel for external use 2.32% tube 100 g
$31.75
Free Worldwide Shipping
to: Australia, Canada, Czechia, Denmark, Estonia, Ireland, Israel, Italy, Japan, Lithuania, Mexico, Netherlands, Poland, Slovakia, South Korea, Switzerland, United Kingdom, United States and more
In stock
Diclosafe Forte gel relieves pain, inflammation and swelling in joints and muscles, treating injuries, rheumatism and osteoarthritis effectively.
Categories: Musculoskeletal system, Pain relief
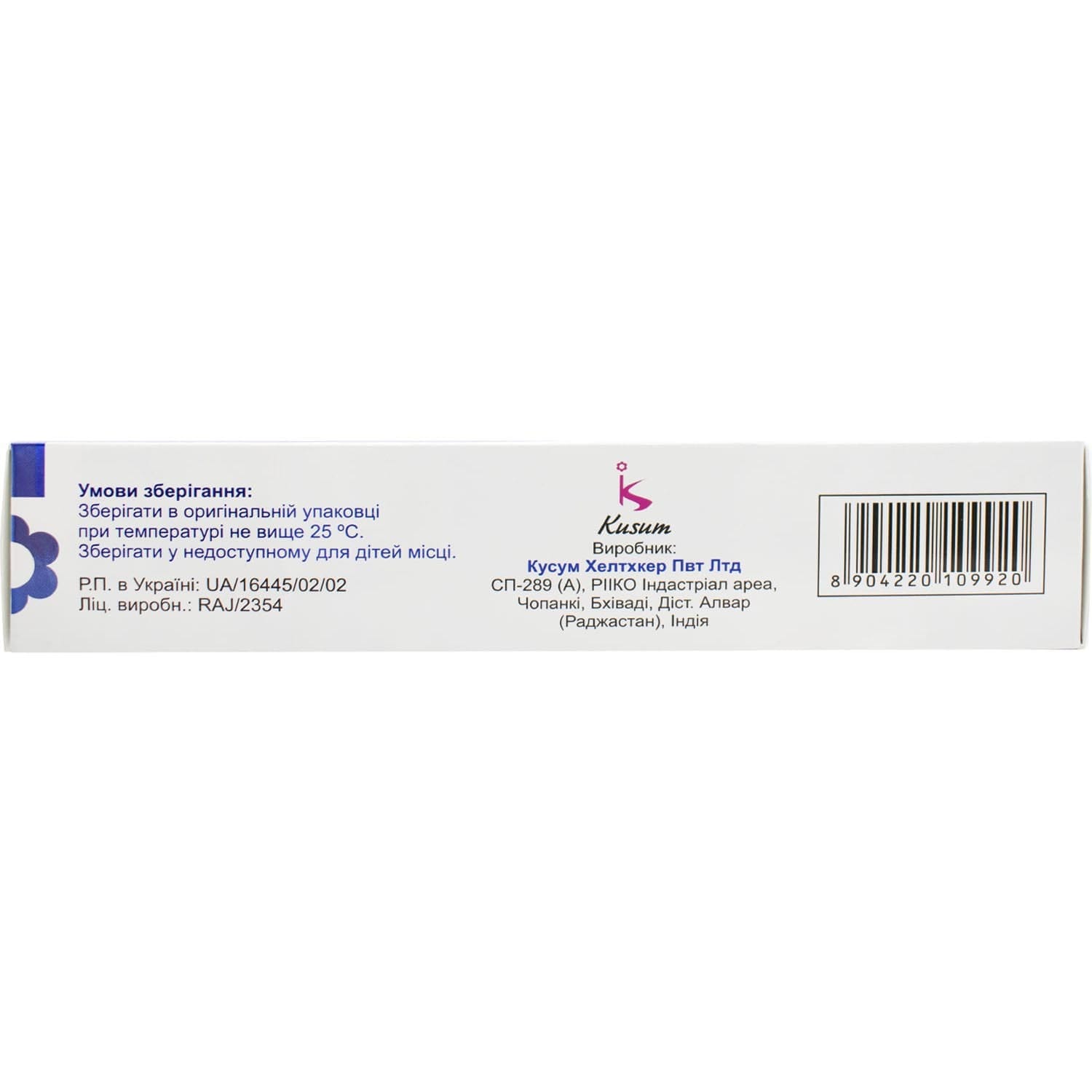
Brand: Kusum Healthcare
Payment
PayPal, Debit or Credit card, Google Pay, Apple Pay
Emulsion gel for external use Diclosafe Forte is indicated for:
- Treatment of pain, inflammation and swelling in:
- soft tissue injuries, injuries to tendons, ligaments, muscles and joints (e.g. due to dislocation, sprain, impact) and back pain (sports injuries)
- localized forms of soft tissue rheumatism: tendonitis (including “tennis elbow”), bursitis, shoulder syndrome and periarthropathy.
- Symptomatic treatment of osteoarthritis of small and medium-sized joints located superficially, such as finger joints or knee joints.
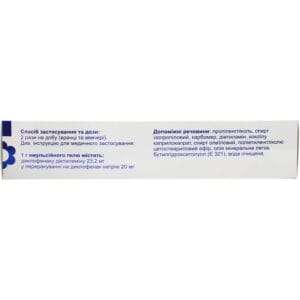
Composition
Active ingredient: diclofenac diethylamine;
1 g of gel contains 23.2 mg of diclofenac diethylamine, equivalent to 20 mg of diclofenac sodium;
Excipients: propylene glycol, isopropyl alcohol, carbomer, diethylamine, cocoyl caprylocaprate, oleyl alcohol, polyethylene glycol cetostearyl ether, light mineral oil, butylhydroxytoluene (E 321), purified water.
Contraindication
- Hypersensitivity to diclofenac, acetylsalicylic acid, other NSAIDs or other components of the drug.
- History of attacks of bronchial asthma, urticaria or acute rhinitis caused by taking acetylsalicylic acid or other NSAIDs.
- Concomitant use with other drugs containing diclofenac.
- Concomitant use with other nonsteroidal anti-inflammatory drugs.
- The last trimester of pregnancy.
Method of application
Adults and children over 14 years: Diclosafe® Forte gel should be applied 2 times a day (morning and evening), lightly rubbing into the skin at the site of pain. The amount of the drug used depends on the size of the affected area (for example, 2-4 g, the size of a cherry or walnut, is enough to apply to an area of 400-800 cm 2).
After applying the drug, hands should be washed, except when this is the area to be treated.
The duration of therapy depends on the nature of the disease and the effectiveness of the treatment.
The drug should not be used for longer than 14 days unless otherwise recommended by a doctor.
You should seek medical advice if the symptoms of the disease do not improve or worsen after 7 days of treatment.
Application features
Pregnant women
By doctor’s prescription.
Children
Since there is insufficient data on the efficacy and safety of diclofenac gel in children under 14 years of age, the drug is contraindicated in children under 14 years of age (see Contraindications). When using the drug in children over 14 years of age for more than 7 days or if the symptoms of the disease worsen, you should seek medical advice.
Drivers
The drug has no or negligible influence on the ability to drive and use machines.
Overdose
Symptoms.
Overdose is unlikely due to the low absorption of diclofenac into the systemic bloodstream when applied topically. At the same time, the development of systemic adverse reactions may occur in case of accidental ingestion of Diclosafe® Forte gel (it should be borne in mind that 1 tube of the drug of 30 g contains the equivalent of 0.6 g of diclofenac sodium). In case of accidental ingestion of the drug and the occurrence of significant systemic adverse reactions, general therapeutic measures used in the treatment of NSAID poisoning should be applied. Gastric lavage and administration of activated charcoal are recommended, especially in cases where the drug has been ingested recently.
Treatment.
Treatment of NSAID overdose consists of supportive and symptomatic therapy. There is no typical clinical picture caused by an overdose of diclofenac gel. Supportive and symptomatic treatment is indicated for complications such as arterial hypotension, renal failure, convulsions, gastrointestinal disorders and respiratory depression. Forced diuresis, hemodialysis or hemoperfusion are unlikely to be useful for the removal of NSAIDs, since the active substances of these drugs are highly bound to plasma proteins and undergo extensive metabolism.
Side effects
Diclofenac gel is usually well tolerated. Adverse reactions include mild, transient skin reactions at the application site. In rare cases, allergic reactions may occur.
Interaction
Since systemic absorption of diclofenac following topical application is very low, the likelihood of interactions is very low. To date, there are no data on interactions with diclofenac when applied topically.
Storage conditions
Store in the original packaging at a temperature not exceeding 25 °C.
Keep out of reach of children.
Shelf life – 2 years.
Be the first to review “Diclosafe Forte emulsion gel for external use 2.32% tube 100 g” Cancel reply
You may also like










Reviews
There are no reviews yet.