No products in the cart.
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇹 Lithuania🇲🇽 Mexico
🇳🇱 Netherlands🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more

Lymphomyosot oral drops droppe...
$36.59 Original price was: $36.59.$33.29Current price is: $33.29.
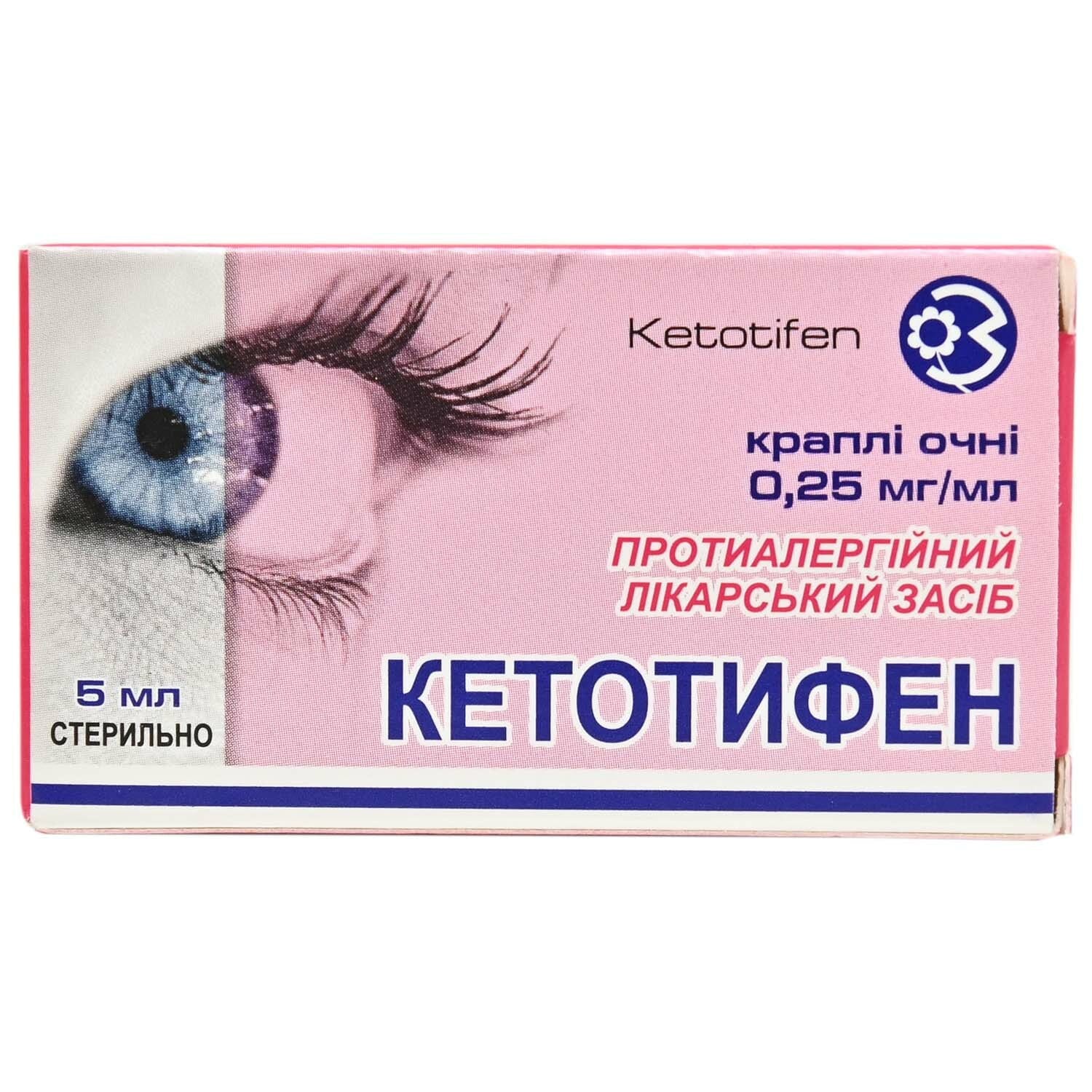
Ketotifen eye drops 0.25 mg/ml in a bottle with a dropper cap 5 ml
$13.83
Free Worldwide Shipping
to: Australia, Canada, Czechia, Denmark, Estonia, Ireland, Israel, Italy, Japan, Lithuania, Mexico, Netherlands, Poland, Slovakia, South Korea, Switzerland, United Kingdom, United States and more
In stock
Ketotifen eye drops relieve and prevent allergic conjunctivitis symptoms like itching, redness and irritation, for seasonal and chronic eye allergies
Categories: Homeopathy, Musculoskeletal system
Brand: BCPP
Payment
PayPal, Debit or Credit card, Google Pay, Apple Pay
Composition and form of release
Active ingredient: ketotifen.
Composition
- 1 ml of solution contains ketotifen fumarate equivalent to ketotifen 0.25 mg;
- Excipients: benzalkonium chloride; sodium chloride; dextran; disodium edetate (Trilon B); sodium hydroxide; citric acid, monohydrate; purified water.
Release form
Eye drops, clear or slightly opalescent, colorless or with a slightly yellowish tinge, liquid.
Indications for use
Prevention and treatment of acute, subacute and chronic allergic conjunctivitis and keratoconjunctivitis, seasonal (spring) conjunctivitis, ocular manifestations of hay fever. In tablet form, it is used to prevent the development of bronchospasm, asthma attacks and acute allergic reactions, to reduce the dose and / or withdrawal of hormonal drugs (corticosteroids) and bronchodilators when used simultaneously.
Application
The drug in the form of drops is prescribed to adults and children over 3 years of age, 1 drop in the conjunctival sac of each eye 2 times a day. Prophylactic administration for subacute and chronic processes should be started 3 weeks before contact with the allergen. Treatment is continued throughout the entire period of allergen exposure. The duration of use of eye drops depends on the type and severity of the disease. The bottle should be carefully closed after each use.
Contraindication
Hypersensitivity to ketotifen and other components of the drug, pregnancy and breastfeeding; children under 3 years of age.
Side effects
Burning and stinging sensation in the eyes, blurred vision immediately after instillation. irritation of the mucous membrane of the eye, skin rash around the eyes; hypersensitivity. When taking the drug orally, drowsiness, dry mouth, dizziness, slowing of mental reactions are possible, which usually disappear after a few days of administration. Occasionally, weight gain is noted, due to increased appetite.
Special instructions
In acute inflammation and during exacerbation of the chronic process, the drug can be used in combination with corticosteroids. The patient should be warned about the need for regular use of the drug. Treatment is started before contact with the allergen and continued throughout the entire period of exposure to the allergen.
The drug should not be used simultaneously with antidiabetic agents.
When using eye drops, soft contact lenses should not be used due to the presence of benzalkonium chloride in the composition. Hard contact lenses should be removed from the eyes immediately before instillation of the drops and inserted no earlier than 15 minutes after application of the drug.
Use of the drug during breastfeeding
Recommended only on doctor’s prescription.
Effects on ability to drive and use machines
When using Pacent tablets, you should refrain from performing potentially hazardous activities that require increased attention. For some time after instilling drops into the eyes, you should refrain from performing work that requires increased attention.
Interactions
With the simultaneous use of ketotifen and antihistamines, a potentiation effect is possible. Simultaneous administration with oral antidiabetic drugs increases the possibility of developing thrombocytopenia.
Overdose
Cases of overdose in the form of drops have not been established. When taken in the form of tablets, confusion and disorientation, tachycardia and decreased blood pressure, convulsions and increased excitability (especially in children), drowsiness and severe sedation, coma are possible. Treatment: gastric lavage, monitoring of cardiovascular system indicators. In case of psychomotor agitation and the development of convulsive syndrome, barbiturates with a short period of action and benzodiazepines are prescribed.
Storage conditions
In a place protected from light at a temperature of 15-25 ° C. Shelf life 2 years. Shelf life of the solution after opening the bottle is 14 days.
| Cyrillic UA | Кетотифен |
|---|---|
| Cyrillic RU | Кетотифен |
Be the first to review “Ketotifen eye drops 0.25 mg/ml in a bottle with a dropper cap 5 ml” Cancel reply
You may also like








Reviews
There are no reviews yet.