No products in the cart.
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇫🇮 Finland
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇻 Latvia
🇱🇹 Lithuania
🇲🇽 Mexico
🇳🇱 Netherlands
🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇫🇮 Finland
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇻 Latvia
🇱🇹 Lithuania
🇲🇽 Mexico
🇳🇱 Netherlands
🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
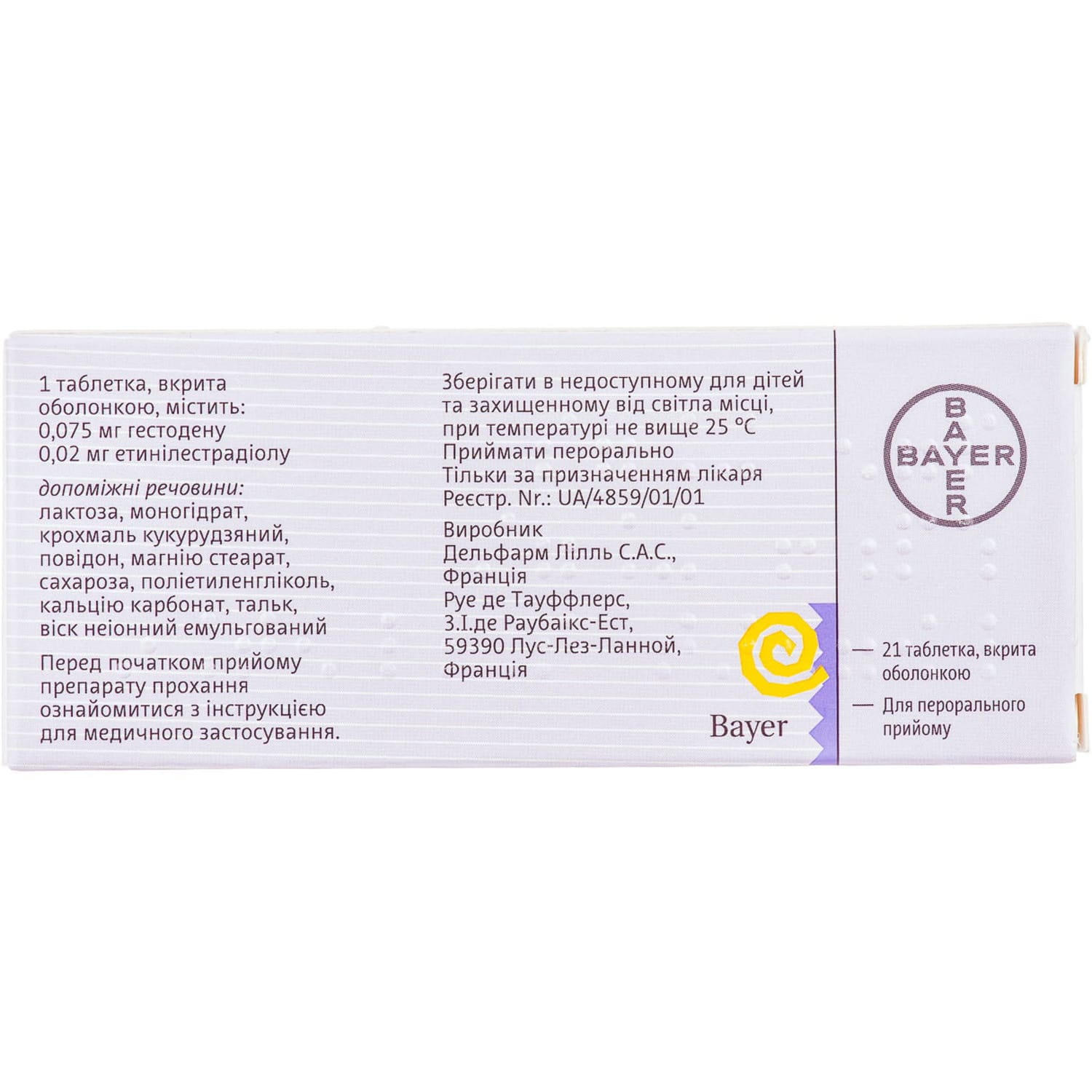
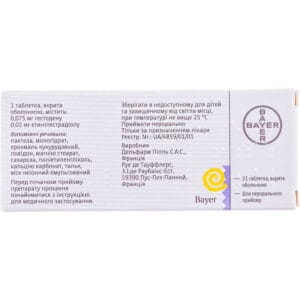
Logest coated tablets blister with calendar scale 21 pcs
$28.24
Free Worldwide Shipping
to: Australia, Canada, Czechia, Denmark, Estonia, Finland, Ireland, Israel, Italy, Japan, Latvia, Lithuania, Mexico, Netherlands, Poland, Slovakia, South Korea, Switzerland, United Kingdom, United States and more
In stock
Logest oral contraceptive tablets with ethinylestradiol and gestodene. Indications, dosage, contraindications, side effects, and safe use details explained.
Categories: Genitourinary system
Brand: Bayer
Payment
PayPal, Debit or Credit card, Google Pay, Apple Pay
Logest tablets are used for oral contraception.
Composition
One film-coated tablet contains (active ingredients):
- ethinylestradiol – 20 mcg;
- gestodena – 75 μg.
Excipients: lactose monohydrate; corn starch; povidone; magnesium stearate; sucrose; polyethylene glycol; calcium carbonate; talc; emulsified nonionic wax.
Contraindication
Combined hormonal contraceptives (CHCs) should not be used if any of the following conditions or diseases are present. If any of these conditions appear for the first time during CHC use, the drug should be discontinued immediately.
Presence or risk of venous thromboembolism (VTE):
- venous thromboembolic events currently (anticoagulant therapy) or in history (e.g. deep vein thrombosis (DVT) or pulmonary embolism (PE));
- known hereditary or acquired predisposition to thromboses, venous thromboembolism (e.g. resistance to activated protein C, including factor V Leiden), antithrombin III deficiency, protein C deficiency, protein S deficiency;
- major surgical interventions with prolonged immobilization;
- high risk of venous thromboembolism due to the presence of multiple risk factors.
Presence or risk of arterial thromboembolism (ATE):
- arterial thromboembolism – the presence of arterial thromboembolism now or in history (for example, myocardial infarction (MI)) or the presence of prodromal conditions (for example, angina);
- cerebrovascular disease – current or history of stroke or presence of prodromal conditions (e.g. transient ischemic attack (TIA));
- known hereditary or acquired predisposition to arterial thromboembolism (e.g. hyperhomocysteinemia) or the presence of antiphospholipid antibodies (anticardiolipin antibodies, lupus anticoagulant);
- migraine with focal neurological symptoms in history;
- high risk of arterial thromboembolism due to the presence of multiple risk factors or the presence of one of the serious risk factors, such as diabetes mellitus with vascular damage, severe arterial hypertension, severe dyslipoproteinemia;
- current or history of severe liver disease, until liver function tests return to normal;
- current or history of liver tumors (benign or malignant);
- known or suspected hormone-dependent malignant tumors (e.g., genital or breast);
- vaginal bleeding of unclear etiology;
- hypersensitivity to the active substances or to any of the components of the drug.
“Logest” is contraindicated for simultaneous use with medications containing St. John’s wort.
The drug “Logest” is contraindicated when used simultaneously with drugs containing ombitasvir / paritaprevir / ritonavir and dasabuvir.
Method of application
Take one tablet once a day regularly at about the same time (with a small amount of liquid if necessary) for 21 consecutive days.
The tablet-taking from each subsequent pack should be started after the seven-day tablet-free interval. Withdrawal bleeding usually begins 2-3 days after taking the last tablet and may continue after starting the next pack.
Application features
Pregnant women
The drug is contraindicated during pregnancy.
The use of Logest is not recommended during breastfeeding, as estrogens / progestins penetrate into breast milk. If a woman wishes to breastfeed, another method of contraception should be chosen.
Children
The drug is indicated for use as prescribed by a doctor only after the onset of regular menstruation.
Overdose
There are no reports of overdose with the drug “Logest”.
Symptoms of overdose with COCs may include nausea, vomiting and withdrawal bleeding. Withdrawal bleeding may occur in girls even before menarche in the event of unintentional/accidental use of the drug. There is no antidote and treatment is symptomatic.
Side effects
Women taking CHCs have been shown to have an increased risk of developing venous or arterial thrombotic/thromboembolic events, including myocardial infarction, stroke, transient ischemic attacks, venous thrombosis and PE.
The most commonly reported adverse reactions (> 10%) in phase III clinical trials and post-marketing experience were headaches, including migraines, and spotting/bleeding.
Storage conditions
Store at a temperature not exceeding 25 °C, out of the reach of children and protected from light.
Shelf life – 3 years.
Be the first to review “Logest coated tablets blister with calendar scale 21 pcs” Cancel reply
You may also like









Reviews
There are no reviews yet.