No products in the cart.
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇫🇮 Finland
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇻 Latvia
🇱🇹 Lithuania
🇲🇽 Mexico
🇳🇱 Netherlands
🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
We deliver to:
🇦🇺 Australia
🇨🇦 Canada
🇨🇿 Czechia
🇩🇰 Denmark
🇪🇪 Estonia
🇫🇮 Finland
🇮🇪 Ireland
🇮🇱 Israel
🇮🇹 Italy
🇯🇵 Japan
🇱🇻 Latvia
🇱🇹 Lithuania
🇲🇽 Mexico
🇳🇱 Netherlands
🇵🇱 Poland
🇸🇰 Slovakia
🇰🇷 South Korea
🇨🇭 Switzerland
🇬🇧 United Kingdom
🇺🇸 United States of America
and more
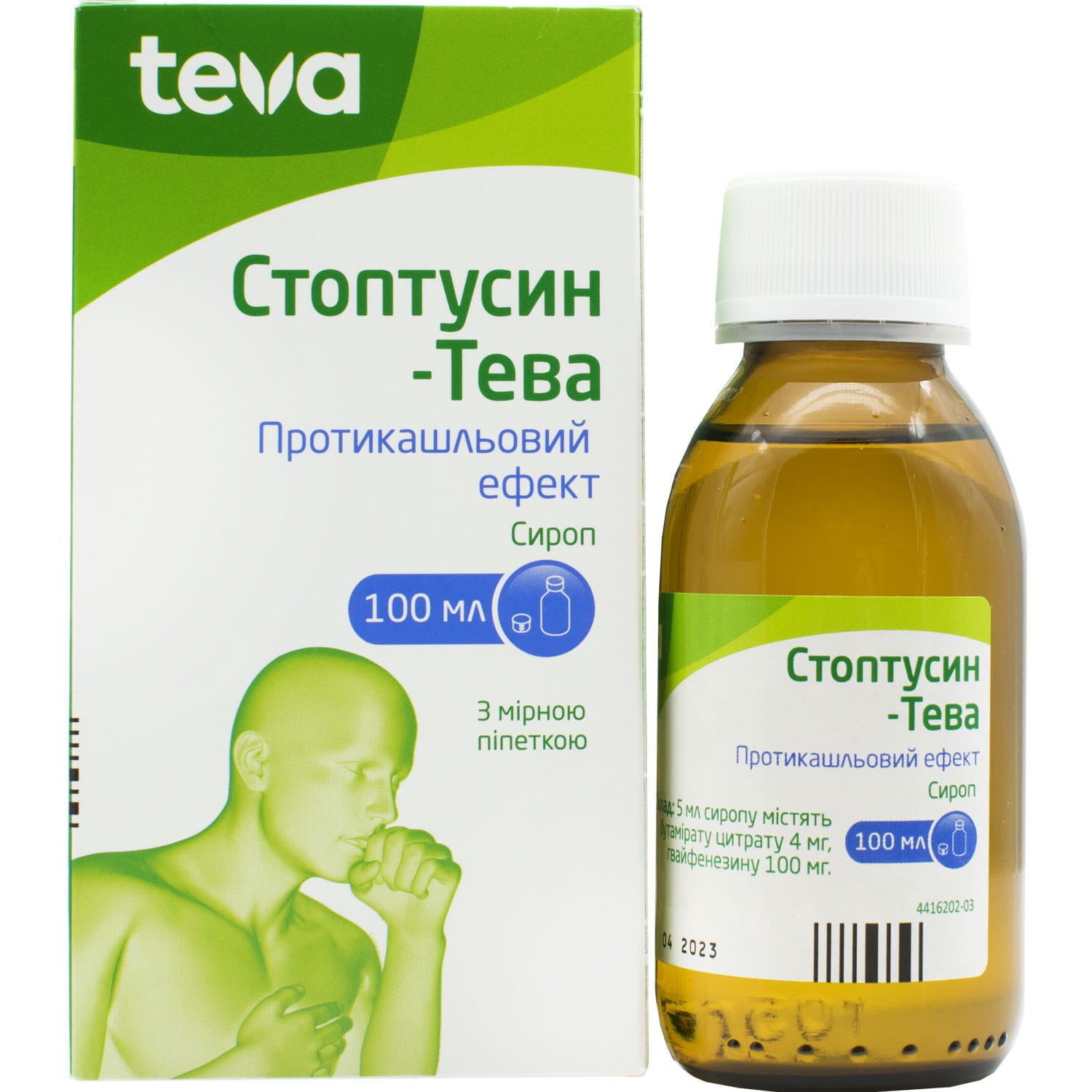
Stoptussin Teva syrup bottle 100 ml
$21.02
Free Worldwide Shipping
to: Australia, Canada, Czechia, Denmark, Estonia, Finland, Ireland, Israel, Italy, Japan, Latvia, Lithuania, Mexico, Netherlands, Poland, Slovakia, South Korea, Switzerland, United Kingdom, United States and more
In stock
Stoptussin combines butamirate and guaifenesin to relieve dry cough, reduce irritation, and support mucus clearance for easier breathing and comfort.
Categories: Cold and flu
Brand: Teva
Payment
PayPal, Debit or Credit card, Google Pay, Apple Pay
Pharmacological properties
Pharmacodynamics. dihydrogen citrate is a combined drug with antitussive, mucolytic and expectorant effects. Butamirate citrate belongs to non-opioid antitussive agents of peripheral action, has a local anesthetic effect on the nerve endings that transmit ascending signals from the respiratory tract. Unlike opioid antitussive agents, it does not cause a central inhibitory effect, does not suppress the respiratory center, and does not cause addiction.
The antitussive effect of butamirate citrate is complemented by the expectorant effect of guaifenesin.
Guaifenesin exhibits secretolytic (by directly increasing the secretion of bronchial glands and stimulating the elimination of acidic glycoproteins from acinar cells) and secretomotor properties (reduces the viscosity of sputum and facilitates the evacuation of mucus and its expectoration).
Pharmacokinetics. Butamirate citrate is rapidly and completely absorbed, 98% binds to plasma proteins. As a result of metabolism, two metabolites are formed that have an antitussive effect, and more than 90% of them are excreted by the kidneys; only a small part is excreted in the feces. Biological T ½ is about 6 hours.
Guaifenesin after oral administration is quickly and easily absorbed in the gastrointestinal tract. A small amount binds to plasma proteins. It is excreted by the kidneys, mainly in the form of metabolites, infrequently – in unchanged form. Biological T ½ – 1 h.
Indication
Dry, irritating, paroxysmal cough of various genesis; the drug in the form of a solution and syrup can be used to eliminate cough in the pre- and postoperative period.
Application
The dose of the drug depends on the patient’s body weight.
Stoptussin tablets: body weight 50 kg – ½ tablet 4 times a day; 50-70 kg – 1 tablet 3 times; 70-90 kg – 1½ tablets 3 times; 90 kg – 2 tablets 3 times a day. The interval between individual doses should be 4-6 hours. The tablet can be divided in half. The drug is recommended to be taken after meals, washed down with a sufficient amount of liquid (water, tea, juice).
Stoptussin drops: body weight 7 kg – 8 drops every 6-8 hours (3-4 times a day); 7-12 kg – 9 drops every 6-8 hours (3-4 times); 13-20 kg – 14 drops every 8 hours (3 times); 21-30 kg – 14 drops every 6-8 hours (3-4 times); 31-40 kg – 16 drops every 6-8 hours (3-4 times); 41-50 kg – 25 drops every 8 hours (3 times); 51-70 kg – 30 drops every 8 hours (3 times); from 71 kg – 40 drops every 8 hours (3 times a day). The drug is recommended to be taken after meals. The appropriate number of drops is diluted in 100 ml of liquid (water, tea or fruit juice). For children who are unable to drink 100 ml of liquid, the amount of liquid can be reduced to ensure the required dose is taken.
The maximum daily dose of guaifenesin for children aged 6 months to 2 years is 300 mg (102 drops), for children aged 2-6 years – 600 mg (204 drops), 6-12 years – 1200 mg/day, for adults and children over 12 years – 2400 mg/day.
Do not take the drug for longer than 7 days without consulting a doctor.
Stoptussin syrup: intended for use in children from 6 months of age and adults. The dosage should be selected depending on the patient’s body weight: up to 12 kg – 1.25 ml 3-4 times a day; 12-20 kg – 2.5 ml 3 times a day; 20-40 kg – 2.5 ml 3-4 times a day; 40-70 kg – 5 ml 3 times a day; 70-90 kg – 5 ml 4 times a day; 90 kg – 7.5 ml 3-4 times a day.
The interval between doses should be 4-6 hours.
It is recommended to take the drug after meals, drinking plenty of fluids (water, tea, fruit juice).
Do not take the drug for longer than 7 days without consulting a doctor.
Contraindication
Hypersensitivity to the components of the drug, myasthenia gravis.
Side effects
If the recommended dosage is followed, patients tolerate the drug well. Side effects usually disappear after reducing the dose.
Metabolic disorders: lack of appetite.
From the nervous system: headache, drowsiness.
Respiratory system: difficulty breathing.
Hearing and vestibular disorders: dizziness.
Gastrointestinal: discomfort, nausea, stomach pain, vomiting, diarrhea. If you experience stomach discomfort or other unusual effects, you should stop using the drug and consult a doctor.
Skin and subcutaneous tissue disorders: allergic reactions, including anaphylactic shock, angioedema, rash, itching, urticaria, rash.
From the urinary system: urolithiasis.
Special instructions
When using the drug, it is necessary to refrain from drinking alcoholic beverages. The drug is not used in patients with:
- productive cough and/or persistent or chronic cough associated with smoking;
- BA;
- chronic bronchitis or emphysema.
The drug is used with caution in asthma, tuberculosis, and pneumococcosis.
If the cough does not go away or worsens, treatment should be reviewed.
The drug in the form of drops contains 35.6 vol.% ethanol (alcohol), i.e. 0.332 g / dose (40 drops), which is equivalent to 8.4 ml of beer or 3.5 ml of wine per dose.
The syrup contains liquid maltitol. Patients with rare hereditary problems of fructose intolerance should not take this medicine.
Pregnancy and lactation: There are no controlled studies in pregnant women in humans or animals.
An increased incidence of inguinal hernia in newborns has been reported with the use of guaifenesin in the first trimester of pregnancy. Therefore, the drug is contraindicated in the first trimester of pregnancy.
The use of the drug in the II and III trimesters of pregnancy or during breastfeeding is possible if, in the opinion of the doctor, the expected positive effect for the mother outweighs the potential risk to the fetus/child.
It is not known whether butamirate citrate or guaifenesin passes into breast milk.
With safety in mind, the benefits and risks of using the drug during breastfeeding should be carefully weighed. The use of the drug during breastfeeding is possible if, in the opinion of the doctor, the expected positive effect for the mother outweighs the potential risk to the child.
Children. The drug in tablet form is used in children from 12 years of age; in the form of drops and syrup – from 6 months according to indications (doses and method of administration are given in the APPLICATION section).
Ability to influence the reaction rate when driving vehicles or working with other mechanisms. In rare cases, Stoptusin causes drowsiness, which may have some effect on the ability to drive vehicles or work with complex mechanisms. Therefore, caution should be exercised when driving vehicles or performing other work that requires attention (for example, when operating mechanisms).
Interactions
Since butamirate suppresses the cough reflex, the simultaneous use of expectorants should be avoided, as this may lead to mucus stagnation in the respiratory tract, which increases the risk of bronchospasm and respiratory tract infection.
Lithium and magnesium enhance the effect of guaifenesin.
Guaifenesin enhances the analgesic effect of paracetamol and acetylsalicylic acid, and also enhances the effect of alcohol and other drugs that suppress the function of the central nervous system. Centrally acting muscle relaxants may increase the severity of side effects of guaifenesin, especially muscle weakness.
Impact on laboratory test results
Guaifenesin may cause false-positive results in diagnostic tests that determine 5-hydroxyindoleacetic acid (photometric method using nitrosonaphthol as a reagent) and vanillylmandelic acid in urine. Therefore, treatment with Stoptussin should be discontinued 48 hours before collecting urine for this analysis.
Overdose
In case of overdose, signs of toxic effects of guaifenesin predominate – drowsiness, muscle weakness, nausea, vomiting, diarrhea, dizziness, arterial hypotension. Radiopaque urolithiasis is possible. There is no specific antidote.
Treatment: gastric lavage, administration of activated charcoal, symptomatic therapy aimed at maintaining the function of the cardiovascular and respiratory systems, kidneys and electrolyte balance.
Storage conditions
In the original packaging at a temperature not exceeding 25 °C. Do not refrigerate or freeze the drops! After opening the bottle, store the syrup for no more than 4 weeks.
Be the first to review “Stoptussin Teva syrup bottle 100 ml” Cancel reply
You may also like











Reviews
There are no reviews yet.